Compare the Balmer series of hydrogen with the series where n, = 4 for the ionized helium atom He*. What is the difference between the wavelengths of the L. (n,, = 5) and L, (n, = 6) line of hydrogen and the n,, = 10 and 12 of He*? (Enter your answers in nm.) - AHe*10 = 433.94 nm - AHe+12 = |410.0702 X nm - He+10 = - AHe*12 nm nm Is there a wavelength of the Balmer series that is very similar (within 0.5 nm) to any wavelength values where n, = 4 in He*? Explain. O Yes, but the wavelengths differ slightly due to different nuclear masses. O No, no wavelength of the Balmer series is within 0.5 nm of a wavelength in the n, = 4 in He* series due to different nuclear masses. O No, no wavelength of the Balmer series is within 0.5 nm of a wavelength in the n, = 4 in Het series due to different nuclear charges. O Yes, but the wavelengths differ slightly due to different nuclear charges.
Compare the Balmer series of hydrogen with the series where n, = 4 for the ionized helium atom He*. What is the difference between the wavelengths of the L. (n,, = 5) and L, (n, = 6) line of hydrogen and the n,, = 10 and 12 of He*? (Enter your answers in nm.) - AHe*10 = 433.94 nm - AHe+12 = |410.0702 X nm - He+10 = - AHe*12 nm nm Is there a wavelength of the Balmer series that is very similar (within 0.5 nm) to any wavelength values where n, = 4 in He*? Explain. O Yes, but the wavelengths differ slightly due to different nuclear masses. O No, no wavelength of the Balmer series is within 0.5 nm of a wavelength in the n, = 4 in He* series due to different nuclear masses. O No, no wavelength of the Balmer series is within 0.5 nm of a wavelength in the n, = 4 in Het series due to different nuclear charges. O Yes, but the wavelengths differ slightly due to different nuclear charges.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter4: Introduction To Quantum Mechanics
Section: Chapter Questions
Problem 59AP
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
PLEASE SHOW ALL STEPS SO I CAN UNDERSTAND HOW TO DO THESE PROBLEMS. THANK YOU IN ADVANCE!
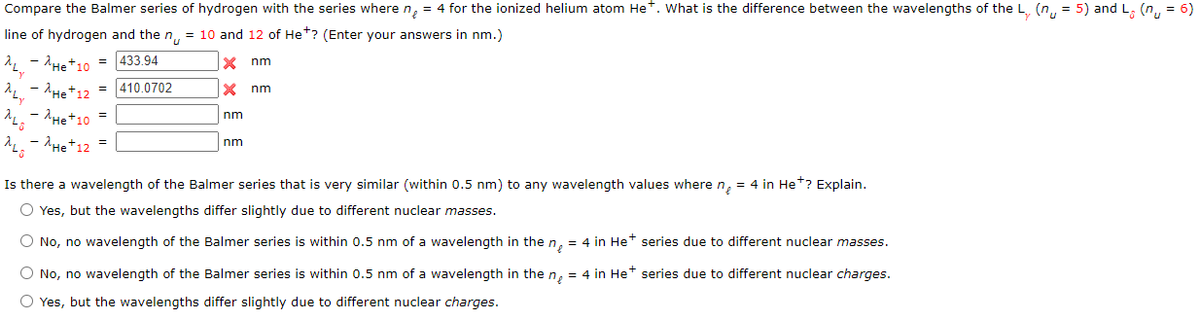
Transcribed Image Text:Compare the Balmer series of hydrogen with the series where n, = 4 for the ionized helium atom Het. What is the difference between the wavelengths of the L. (n, = 5) and L, (n,, = 6)
line of hydrogen and the n, = 10 and 12 of He*? (Enter your answers in nm.)
- AHet10 = 433.94
nm
- 'He+12 = |410.0702
He+10
nm
nm
- AHe+12 =
nm
Is there a wavelength of the Balmer series that is very similar (within 0.5 nm) to any wavelength values wheren, = 4 in Het? Explain.
O Yes, but the wavelengths differ slightly due to different nuclear masses.
O No, no wavelength of the Balmer series is within 0.5 nm of a wavelength in the n, = 4 in Het series due to different nuclear masses.
O No, no wavelength of the Balmer series is within 0.5 nm of a wavelength in the n, = 4 in Het series due to different nuclear charges.
O Yes, but the wavelengths differ slightly due to different nuclear charges.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
