Compare the solubility of nickel(II) sulfide in each of the following aqueous solutions: Clear All 0.10 M Ni(CH3COO)2 More soluble than in pure water. 0.10 M K2S Similar solubility as in pure water. 0.10 M KCH3COO Less soluble than in pure water. 0.10 M NANO3
Compare the solubility of nickel(II) sulfide in each of the following aqueous solutions: Clear All 0.10 M Ni(CH3COO)2 More soluble than in pure water. 0.10 M K2S Similar solubility as in pure water. 0.10 M KCH3COO Less soluble than in pure water. 0.10 M NANO3
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter27: Relative Stabilities Of Complex Ions And Precipitates Prepared From Solutions Of Copper(ii)
Section: Chapter Questions
Problem 1ASA
Related questions
Question
I need hand written solution only
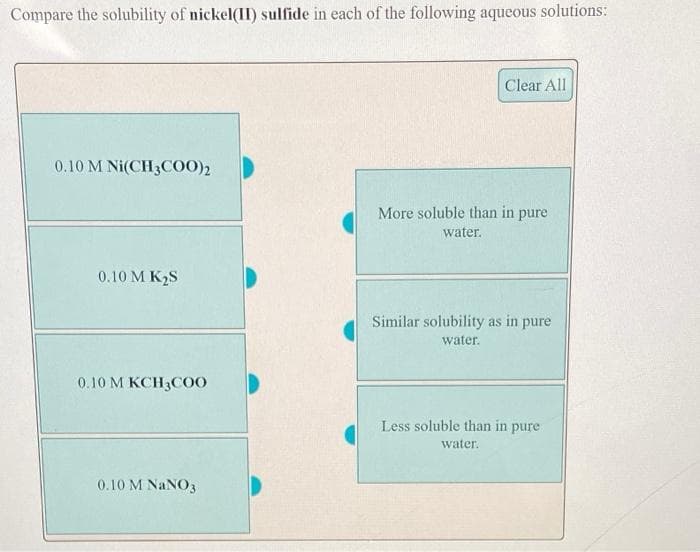
Transcribed Image Text:Compare the solubility of nickel(II) sulfide in each of the following aqueous solutions:
Clear All
0.10 M Ni(CH3CO)2
More soluble than in pure
water.
0.10 M K2S
Similar solubility as in pure
water.
0.10 M KCH3C00
Less soluble than in pure
water.
0.10 M NANO3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole