Complete table 2. * Since you were not able to conduct an actual experiment on these tests, kindly get the information needed to complete the table from textbooks, research articles or other A. references. Tests Principle behind functional group that will evidence of a References the test give a positive result positive result You may draw the reaction Bromine test Acrolein test Lieberman- Burchard test
Complete table 2. * Since you were not able to conduct an actual experiment on these tests, kindly get the information needed to complete the table from textbooks, research articles or other A. references. Tests Principle behind functional group that will evidence of a References the test give a positive result positive result You may draw the reaction Bromine test Acrolein test Lieberman- Burchard test
Basic Clinical Laboratory Techniques 6E
6th Edition
ISBN:9781133893943
Author:ESTRIDGE
Publisher:ESTRIDGE
Chapter6: Basic Clinical Chemistry
Section6.8: Fecal Occult Blood Test
Problem 1CS
Related questions
Question
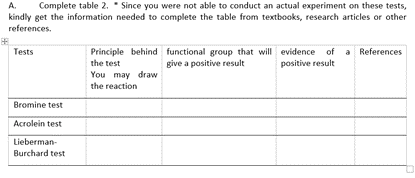
Transcribed Image Text:Complete table 2. * Since you were not able to conduct an actual experiment on these tests,
kindly get the information needed to complete the table from textbooks, research articles or other
A.
references.
Tests
Principle behind functional group that will evidence of a References
the test
give a positive result
positive result
You may draw
the reaction
Bromine test
Acrolein test
Lieberman-
Burchard test
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 5 images

Recommended textbooks for you





