Complete the half reaction ZnS(s) → Zn(s) + S²^(aq) O a) ZnS(s) + 1e → Zn(s) + S²(aq) s2-(aq) b) ZnS(s) → Zn(s) + S²-(aq) + 1e c) ZnS(s) + 2e Zn(s) + s2-(aq) O d) ZnS(s) –→ Zn(s) + S²´(aq) + 2e¯
Complete the half reaction ZnS(s) → Zn(s) + S²^(aq) O a) ZnS(s) + 1e → Zn(s) + S²(aq) s2-(aq) b) ZnS(s) → Zn(s) + S²-(aq) + 1e c) ZnS(s) + 2e Zn(s) + s2-(aq) O d) ZnS(s) –→ Zn(s) + S²´(aq) + 2e¯
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter17: Electrochemistry And Its Applications
Section: Chapter Questions
Problem 66QRT
Related questions
Question
Please help me with this and explain it it first
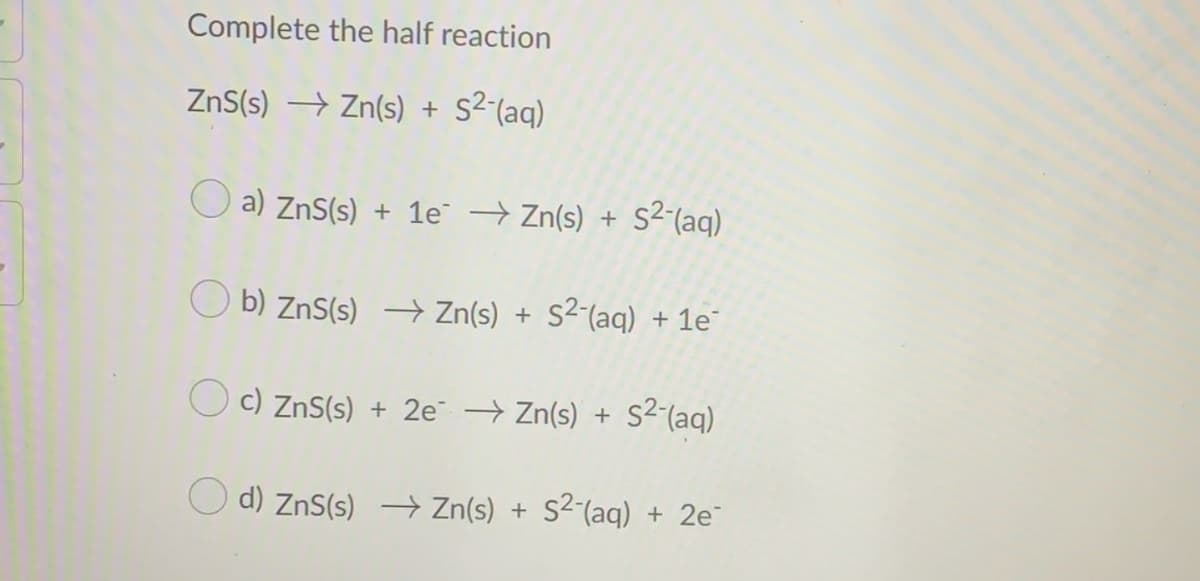
Transcribed Image Text:Complete the half reaction
ZnS(s) → Zn(s) +
s2 (aq)
O a) ZnS(s) + 1e → Zn(s) + S2(aq)
O b) ZnS(s) → Zn(s) + S²-(aq) + 1e
O c) ZnS(s) + 2e Zn(s) + s2(aq)
d) ZnS(s) → Zn(s) + S²´(aq) + 2e
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning