Complete the spectroscopy data tables for a compound with molecular formula C3H5BrO2. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular 1H NMR spectru
Complete the spectroscopy data tables for a compound with molecular formula C3H5BrO2. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular 1H NMR spectru
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter16: Introduction To Magnetic Spectroscopy
Section: Chapter Questions
Problem 16.41E
Related questions
Question
Complete the spectroscopy data tables for a compound with molecular formula C3H5BrO2.
Determine the structure of the compound. Any labile protons, if they exist, will not be
present in this particular 1H NMR spectrum.
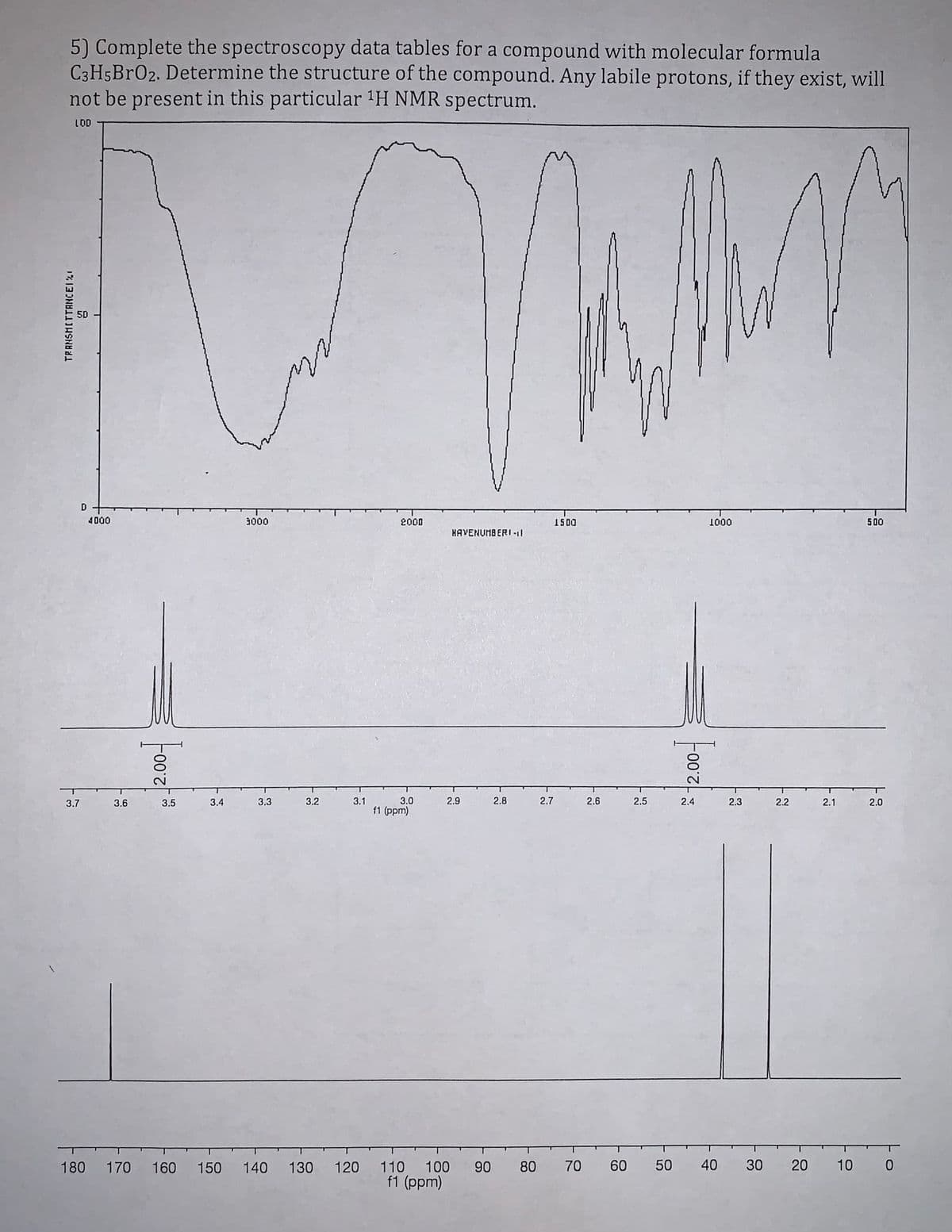
Transcribed Image Text:5) Complete the spectroscopy data tables for a compound with molecular formula
C3H5BR02. Determine the structure of the compound. Any labile protons, if they exist, will
not be present in this particular 'H NMR spectrum.
4000
3000
2000
15 0O
1000
500
HAVENUMBERI-|
2.7
3.1
f1 (ppm)
3.7
3.6
3.5
3.4
3.3
3.2
3.0
2.9
2.8
2.6
2.5
2.4
2.3
2.2
2.1
2.0
130 120 110 100 90
f1 (ppm)
180 170 160 150 140
80
70
60
40
10
20
30
12.00-
50
00-
TPANSHITTANCEI%I
![5- Degrees of Unsaturation (show work):
DOU = 1
C3 Hg Br02
[2(3)+2-5 +0-]
2.
IR Data Table
13C NMR Data Table
13
Stretch (cm)
Bond
13
C Signal Chemical Shift (ppm) PC Signal
1
171ppm
पि्सरी
2.
38
29
'H NMR Data Table
H Signal
Chemical Shift (ppm) | Integration
Multiplicity
3.525
2H
t
Proposed Structure
2.4
2H
t](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F41ea191e-828c-4f9d-b6fb-1ac8fc340597%2F5ddd1b35-8e43-45b4-98cc-e3da74c600b9%2Fxm82fy_processed.jpeg&w=3840&q=75)
Transcribed Image Text:5- Degrees of Unsaturation (show work):
DOU = 1
C3 Hg Br02
[2(3)+2-5 +0-]
2.
IR Data Table
13C NMR Data Table
13
Stretch (cm)
Bond
13
C Signal Chemical Shift (ppm) PC Signal
1
171ppm
पि्सरी
2.
38
29
'H NMR Data Table
H Signal
Chemical Shift (ppm) | Integration
Multiplicity
3.525
2H
t
Proposed Structure
2.4
2H
t
Expert Solution
Step 1
IR Data:
3100-3000cm-1 = -OH stretch, broad
1700 cm-1 = -C=O group
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,