Complete the table below by deciding whether a precipitate forms w aqueous solutions A and B are mixed. If a precipitate will form, ent empirical formula in the last column. Does a precipitate form when A and B empirical formula of solution A solution B precipitate are mixed? sodium chloride ammonium nitrate yes no zinc chloride ammonium sulfide yes no iele blenid
Complete the table below by deciding whether a precipitate forms w aqueous solutions A and B are mixed. If a precipitate will form, ent empirical formula in the last column. Does a precipitate form when A and B empirical formula of solution A solution B precipitate are mixed? sodium chloride ammonium nitrate yes no zinc chloride ammonium sulfide yes no iele blenid
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter16: Solubility And Complex Ion Equilibria
Section: Chapter Questions
Problem 98AE: In the chapter discussion of precipitate formation, we ran the precipitation reaction to completion...
Related questions
Question
100%
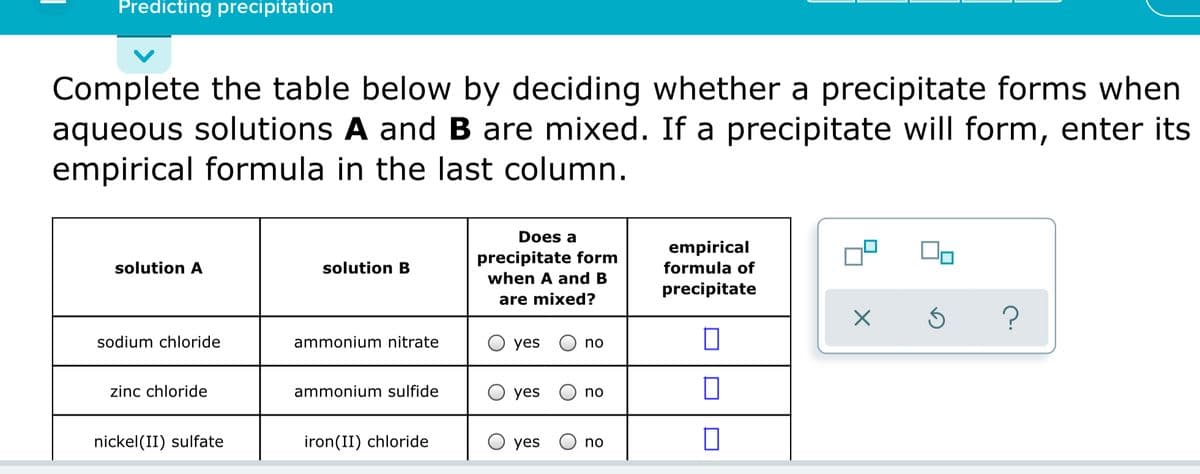
Transcribed Image Text:Predicting precipitation
Complete the table below by deciding whether a precipitate forms when
aqueous solutions A and B are mixed. If a precipitate will form, enter its
empirical formula in the last column.
Does a
empirical
precipitate form
solution A
solution B
formula of
when A and B
precipitate
are mixed?
sodium chloride
ammonium nitrate
yes
no
zinc chloride
ammonium sulfide
yes
no
nickel(II) sulfate
iron(II) chloride
yes
no
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning