Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are mixed. If a precipitate will form, enter its empin formula in the last column. Does a precipitate form when A and B are mixed? empirical formula of precipitate solution A solution B iron(III) nitrate potassium hydroxide yes O no lead(II) nitrate sodium sulfide O yes O no sodium hydroxide ammonium nitrate O yes O no
Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are mixed. If a precipitate will form, enter its empin formula in the last column. Does a precipitate form when A and B are mixed? empirical formula of precipitate solution A solution B iron(III) nitrate potassium hydroxide yes O no lead(II) nitrate sodium sulfide O yes O no sodium hydroxide ammonium nitrate O yes O no
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 39P
Related questions
Question
This is not a graded question
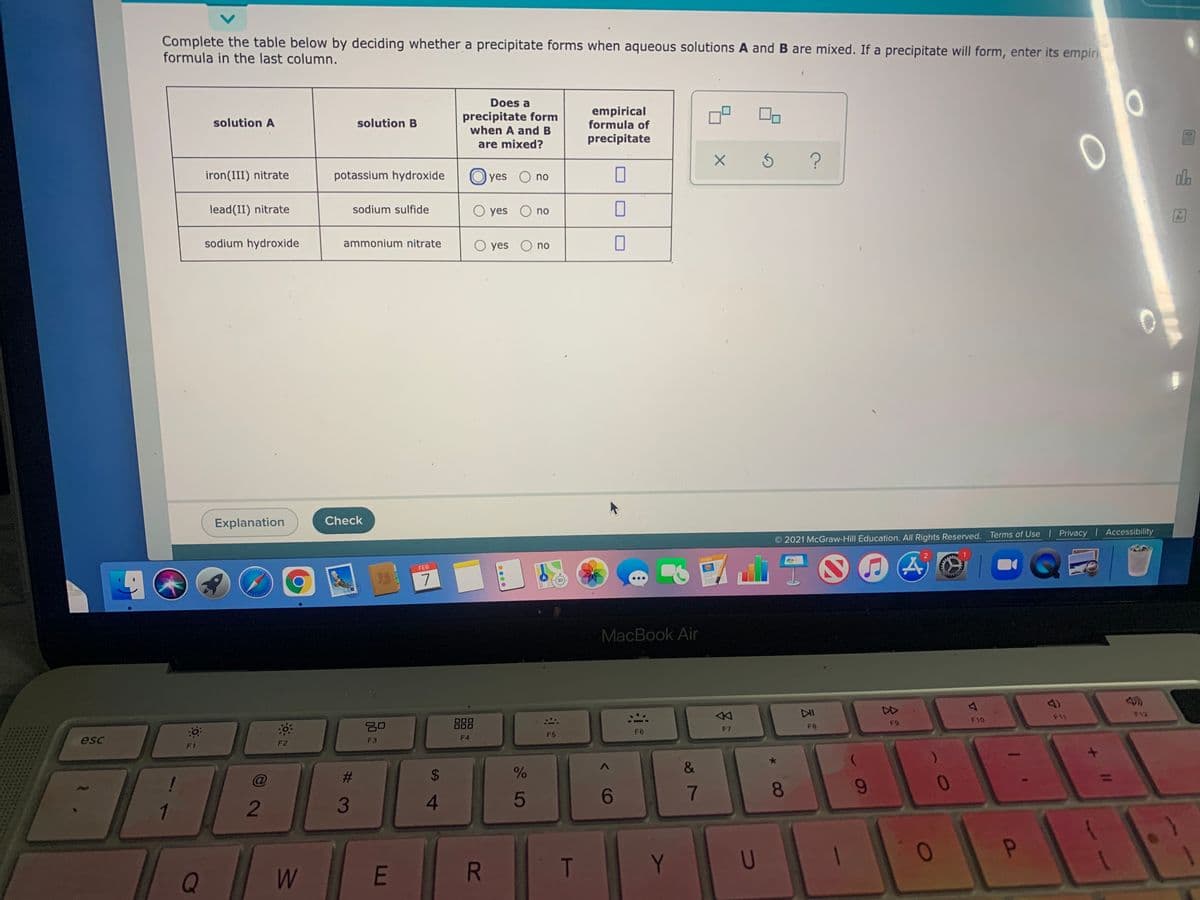
Transcribed Image Text:Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are mixed. If a precipitate will form, enter its empiri
formula in the last column.
Does a
precipitate form
when A and B
empirical
formula of
solution A
solution B
are mixed?
precipitate
iron(III) nitrate
potassium hydroxide
yes O no
do
lead(II) nitrate
sodium sulfide
O yes O no
sodium hydroxide
ammonium nitrate
O yes O no
Explanation
Check
O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use | Privacy Accessibility
PAGES
2
FEB
7
MacBook Air
4)
888
DII
esc
F8
F10
911
12
F2
F3
F4
F5
F6
F7
F1
23
24
1
3
5
8.
9.
01
T
U
1
P.
会。
87
R
W/
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning
