Complete the table by calculating the reaction quotient and determining the direction of these reactions: (A) A 1.00-L flask containing 0.0500 mol of NO(g), 0.0155 mol of Cla(g), and 0.500 mol of NOCI: 2NO(g) + Cla(g) = 2NOCI(g) (B) A 5.0-L flask containing 17 g of NH3, 14 g of N2, and 12 g of H2: N2 (g) + 3H2(g) = 2NH3(g) (C) A 2.00-L flask containing 230 g of SO3(g): 2S03(g) = 2s02(g) + 02(g) Reaction Kc Values Reaction Direction of Reaction Quotient (Forward or Reverse ) A 4.6x104 B 0.060 0.230
Complete the table by calculating the reaction quotient and determining the direction of these reactions: (A) A 1.00-L flask containing 0.0500 mol of NO(g), 0.0155 mol of Cla(g), and 0.500 mol of NOCI: 2NO(g) + Cla(g) = 2NOCI(g) (B) A 5.0-L flask containing 17 g of NH3, 14 g of N2, and 12 g of H2: N2 (g) + 3H2(g) = 2NH3(g) (C) A 2.00-L flask containing 230 g of SO3(g): 2S03(g) = 2s02(g) + 02(g) Reaction Kc Values Reaction Direction of Reaction Quotient (Forward or Reverse ) A 4.6x104 B 0.060 0.230
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter13: Fundamental Equilibrium Concepts
Section: Chapter Questions
Problem 12E: Show that the complete chemical equation, the total ionic equation, and the net ionic equation for...
Related questions
Question
No need for solutions, just the correct answer. Thank you!
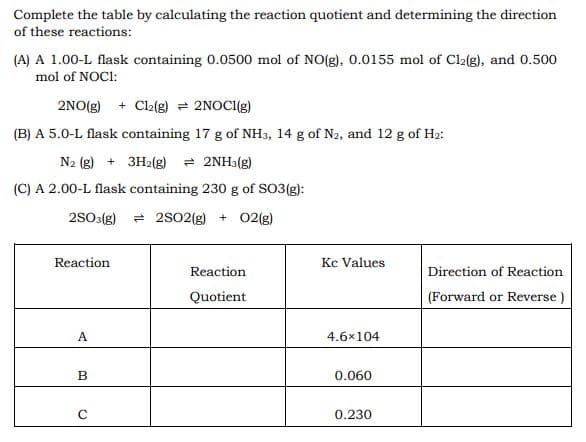
Transcribed Image Text:Complete the table by calculating the reaction quotient and determining the direction
of these reactions:
(A) A 1.00-L flask containing 0.0500 mol of NO(g), 0.0155 mol of Cla(g), and 0.500
mol of NOCI:
2NO(g) + Cl2(g) = 2NOCI(g)
(B) A 5.0-L flask containing 17 g of NH3, 14 g of N2, and 12 g of H2:
N2 (g) + 3H2(g) = 2NH3(g)
(C) A 2.00-L flask containing 230 g of SO3(g):
2S0:(g)
= 2s02(g) + 02(g)
Reaction
Kc Values
Reaction
Direction of Reaction
Quotient
(Forward or Reverse )
A
4.6x104
B
0.060
0.230
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning