Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter20: Organic Chemistry
Section: Chapter Questions
Problem 7QAP
Related questions
Question
Explain following question ❓
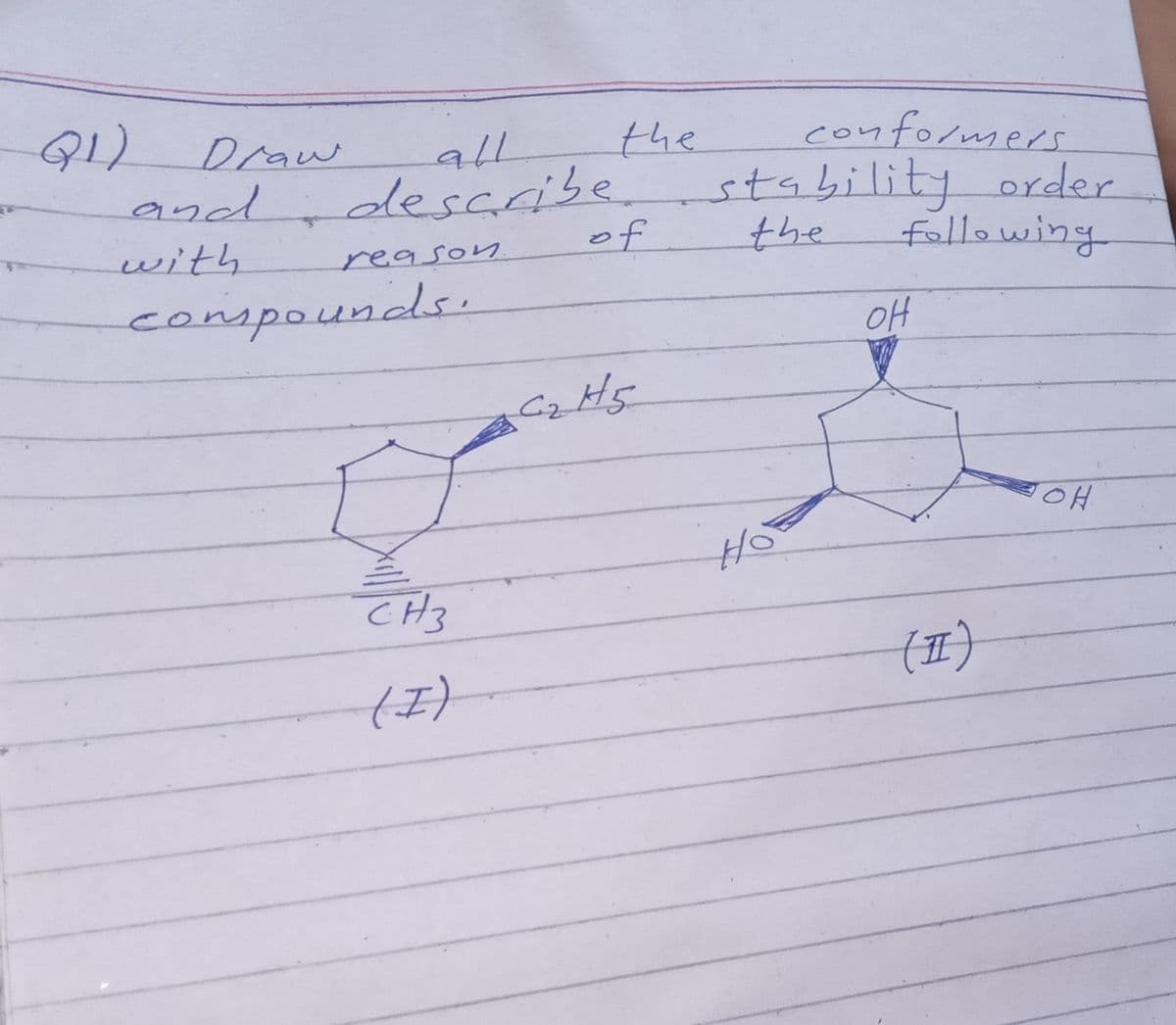
Transcribed Image Text:1)
conformers
the
stability order
the
Draw
all
and describe
with
of
fllowing
reason
compounds.
oH
Cz
HO.
CH3
(표)
()
Expert Solution
Step 1
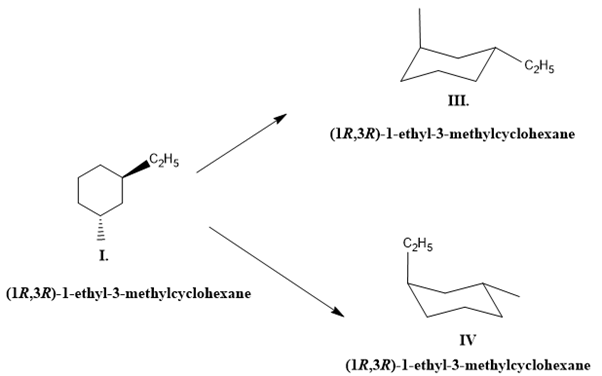
For the given compound (I), the possible conformations are shown by (III) and (IV). Form the III and IV, III is more stable than the IV because the larger group, ethyl, is at an equatorial position.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning