Consider a particular gas. The isothermal compressibility and coefficient of thermal expansion are given by: V K = a =+2V, where c, and c, are numerical constants. C1
Q: If you have 0.411 m³ of water at 25.0 °C in an insulated container and add 0.117 m³ of water at 95.0…
A: The heat content of a system remains conserved. If one component releases heat, other will absorb…
Q: A chemical reaction takes place in a container fitted with a piston of cross-sectional area 75.0…
A: The work done by the system is calculated by the formula as follow:
Q: A sample of helium (perfect gas) undergoes a following two-step process.1. Isothermal reversible…
A: According to the first law of thermodynamics, A) For a reversible isothermal process, the…
Q: What is the final temperature of a 1.00 mol sample of helium gas were it undergoes to a reversible…
A: Given: n = 1.00 mol of helium gas It undergoes to a reversible adiabatic compression V1 = 62.4 L…
Q: A sample of nitrogen initially at T = 25 oC and p = 4.0 atm triples its initial volume. What will be…
A:
Q: Calculate V−1(∂V/∂T)p,n for an ideal gas?
A: Ideal gas- An ideal gas is a theoretical gas composed of many randomly moving point particles that…
Q: A rubberized, cubic chamber was filled with butane gas and was expanded from 3.0L to 4.5L at a…
A:
Q: What will be the molar heat capacity at constant pressure of the gas when 229 J of energy is…
A:
Q: 1 mol N2 gas with a volume of 300 K and 2 L undergoes an isothermal reversible expansion to the…
A: Given that: number of moles of gas = 1 mole temperature = 300 K initial volume, Vi = 2 L final…
Q: 633 mmHg and 289 K Express your answer with the appropriate units. V= 0.65 atm and 77 ∘C Express…
A:
Q: One mole of gas expands isothermically to double its volume. If the gas temperature is 340 K, what…
A:
Q: Calculate the final temperature and the change in enthalpy when 1.15 kJ of energy is transferred as…
A: The heat transferred at constant pressure (qp) is equal to the change in enthalpy (ΔH) of the…
Q: Proof that: a²TV Ср — Су K Where k is the isothermal compressibility
A:
Q: Question attached
A: Given data : Volume (V) = 1m3 Pressure (P) = 345 kPa Temperature (T1) = 20°C = 273 + 20 = 293 K
Q: The temperature change associated with the adiabatic expansion of an ideal gas is defined as: Trinal…
A: The given process is of adiabatic expansion in which expansion of gas take place without any heat…
Q: is the relationship between pressure and volume in the adiabatic processes of an ideal gas, where r…
A: In this equation, we have to use the short form of the ideal gas equation PV=nRT
Q: What is Isothermal Expansion of a van der Waals Gas?
A:
Q: Internal pressure is roughly equal to zero at high temperatures.
A: Internet pressure is used to measure the internet energy of the system.
Q: A sample of 4.0 mol oxygen gas is originally confined in 20 cubic decimeter at 270k and then…
A: Given data,n=4.0molInitial volume=20dm3Final volume=3×20=60dm3P=600torrCp for Oxygen at 270oC=29.355…
Q: Use the Maxwell relations to express the derivatives () (O. H) and (). in terms of the heat…
A: the partial derivative of the thermodynamic variables are related to each other
Q: One mole sample of CO2(g) occupies 2.00 dm3 at a temperature of 300K. If the gas is compressed…
A: The van der Waals equation is given as:
Q: When 178 J of energy is supplied as heat at constant pressure to 1.9 mol of gas molecules, the…
A: Given values: Energy supplied = 178 Joule moles of gas = 1.9 moles Temperature increase = 1.78 K
Q: Which of the following is a measure of the fractional change in volume when the pressure is…
A: In an isothermal process, the temperature is constant.
Q: A supersonic nozzle is a cone-shaped object with a smallhole in the end through which a gas is…
A: The average speed is the mean of the speeds of different molecules of the gas. It is related to the…
Q: It is the relationship between pressure and volume in the adiabatic processes of an ideal gas, where…
A: PV relation for adiabatic process of Ideal gas is
Q: A chemical reaction takes place in a container of cross-sectional area 100cm^2. As a result of the…
A:
Q: What is the final temeprautre of a monoatomic ideal gas that is compressed reversible and…
A: We know that for adiabatic reversible process : TV^v-1 = constant Or, T1V1^v-1 = T2V2^v-1 Or,…
Q: What fraction of the water vaporization enthalpy is consumed in the expansion of water vapor?
A: Answer
Q: 9. Consider a sample of water vapour, which expands reversibly and adiabatically from 97.3 Torr and…
A: We have reversible adiabatic expansion of water from 97.3 torr 400 cm3 to 5 dm3 , we have to…
Q: Investigate the dependence of pV on V for real gases.
A: In case of ideal gas product of pressure and volume in isothermal process remains constant as: for…
Q: Calculate the final pressure of a sample of water vapour that expands reversibly and adiabatically…
A: Adiabatic process are those where there is no exchange of heat. The expression followed by ideal…
Q: 4. Suppose you have 5 moles of helium gas (ideal, monatomic), which expands under constant pressure.…
A:
Q: Use the tables of moist-air properties to find the enthalpy and specific volume for air at 125°F…
A:
Q: How much work is done in blowing up a balloon from zero volume to a volume of 2.2 L, assuming that p…
A: In a system when volume changes then we can say there may some work is done . Given that, V1= 0 L…
Q: A sample of 2.2 mol CO2(g) is originally confined in 15 dm3 at 280 K and then undergoes adiabatic…
A:
Q: A chemical reaction takes place in a container of cross-sectional area 50.0 cm². As a result of the…
A:
Q: he heat capacities of nitrogen gas at constant volumes were measured at the ollowing temperatures…
A: a) Step 1:Make a new Excel file and put the datas as shown T(K) CV(J .mol-1.K-1) 300 20.8 400…
Q: Two ideal gas systems undergo reversible expansion under different conditions starting from the same…
A:
Q: Everything needed is included Consider the reversible and isothermal compression of 2.0 moles of…
A: A reversible and isothermal compression process is considered. In this process, the change in…
Q: One kg of air occupies 0.048 m at 12.5 bar and 537 C° it is expanded at constant temperature to a…
A: Ideal gas equation is given by, PV=nRT where P is the pressure, V is the volume and T is the…
Q: a) Show that), (KrP – BT)V KT = Isothermal compressibility B = volume expansivity
A: dU = TdS - PdVDifferentiaite both sides with respect to P at constant TSo, ∂U∂PT = T∂S∂PT - P∂V∂PT…
Q: One mole of He gas with CV,m = 3R/2 essentially independent of temperature expands reversibly from…
A: Cv,m = 3/2 R => Cp,m = Cv,m + R = 3/2R + R = 5/2R => γ = Cp,m/Cv,m = 5/3 For reversible…
Q: Calculate AG (cal) for the isothermal compression of 66.4 gram of water from 44.2 atm to 116.2 atm…
A:
Q: Carbon Dioxide has a critical temperature of 304.13 K, criticai pressure of 7377.3 kPa and critical…
A: Critical temperature is defined as the temperature at which a susbtance can exist as liquid.…
Q: For a van der Waals gas: a Calculate AUm (in / mol-) for the isothermal reversible expansion of N2…
A:
What are the appropriate units for c1 and c2?
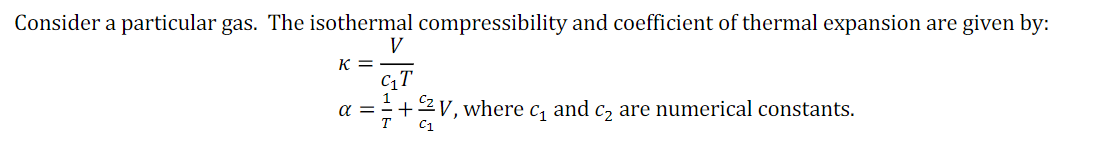
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- The virial equation of state may also be written as an expansion in terms of pressure: Z = 1 + B'p + ... The critical constants for water, H2O, are 218.3 atm. 55.3 cm3 mol-1 and 647.4 K. Assuming that the expansion may be truncated after the second term, calculate the value of the second virial coefficientB' at the critical temperature.Calculate the isothermal compressibility and isobaric thermal volume expansion from Virial equation in terms of pressure. Virial Equation in terms of pressure: PV = A + Bp + Cp2 + Dp3 + ...Calculate the final pressure of a sample of carbon dioxide that expands reversibly and adiabatically from 67.4 kPa and 0.50 dm3 to a final volume of 2.00 dm3. Take γ = 1.4.
- A sample of helium (perfect gas) undergoes a following two-step process.1. Isothermal reversible expansion state 1 (p = 3.0 atm, V = 10.0 L T = 300K) to state 2 (V = 30 L))2. Isobaric compression from state 2 to state 3 (V = 10.0 L) A) What is w and q during step 1? B) What is w and q during step 2? C) What is delta U for the whole process? E) What is delta H for the whole process?Calculate the final pressure of a sample of water vapour that expands reversibly and adiabatically from 87.3 Torr and 500 cm3 to a final volume of 3.0 dm3. Take (gamma) γ = 1.3. ANs in t0rrA sample of nitrogen initially at T = 25 oC and p = 4.0 atm triples its initial volume. What will be its final temperature and pressure if it expands reversibly adiabatically?
- An automobile wheel contains air with a pressure of 3x10^5 Pa at 25 ° C. The sipop cover was removed and the air was expanded adiabatically against an external pressure of 105 Pa. What is the final temperature of the gas in the wheel? (It should be taken into account that the gas behaves ideally and the Cp value for air is 7 / 2R)1 kg of benzene is poured into a flask and heated to 353 K (the boiling point of benzeneat 1 atm.) using a heating element that has a power of 700 W (we ignore thatit's probably not so healthy and smart to do). After 5 minutes, when the benzene has stoodboiling point, the heating element is taken up, and the remaining mass of benzene in the flaskmeasured at 470 grams. Determine the enthalpy of vaporization of benzene.7. Two vessels A and B of different sizes are connected by a pipe which valve. A pipe with a valve. Vessel A contains 142 L of air at 2,767.92 kPa, 93.33 OC. Vessel B, of unknown volume, contains air at 68.95 kPa, 4.44 OC. The valve is opened and, when the properties have been determined, it is found that the pressure and temperature of the mixture of gasses is 1378.96 kPa, and 43.33 OC. What is the volume of vessel B. Use R = 287.08 Joules/Kg-KO for Air

