Consider the chemical equation for the combustion of ammonia: 4NH;(g) + 70,(g) – 4NO,(g) + 6H,0(g) Which statement provides the correct and standard interpretation of the chemical equation? 12 pounds of NH3(g) react with 14 pounds of O,(g) to produce 8 pounds of NO;(g) and 6 pounds of H20(g). 4 mol of NH3(g) react with 7 mol of O,(g) to produce 4 mol of NO,(g) and 6 mol of H,0(g) 89.6 g of NH3(g) react with 156.8 g of O,(g) to produce 89.6 g of NO2(g) and 134.4 g of H2O(g). 22.4 L of NH3(g) react with 22.4 L of O,(g) to produce 22.4 L of NO,(g) and 22.4 L of H2O(g).
Consider the chemical equation for the combustion of ammonia: 4NH;(g) + 70,(g) – 4NO,(g) + 6H,0(g) Which statement provides the correct and standard interpretation of the chemical equation? 12 pounds of NH3(g) react with 14 pounds of O,(g) to produce 8 pounds of NO;(g) and 6 pounds of H20(g). 4 mol of NH3(g) react with 7 mol of O,(g) to produce 4 mol of NO,(g) and 6 mol of H,0(g) 89.6 g of NH3(g) react with 156.8 g of O,(g) to produce 89.6 g of NO2(g) and 134.4 g of H2O(g). 22.4 L of NH3(g) react with 22.4 L of O,(g) to produce 22.4 L of NO,(g) and 22.4 L of H2O(g).
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter3: Stoichiometry
Section: Chapter Questions
Problem 7RQ: Consider the hypothetical reaction between A2 and AB pictured below. What is the balanced equation?...
Related questions
Question
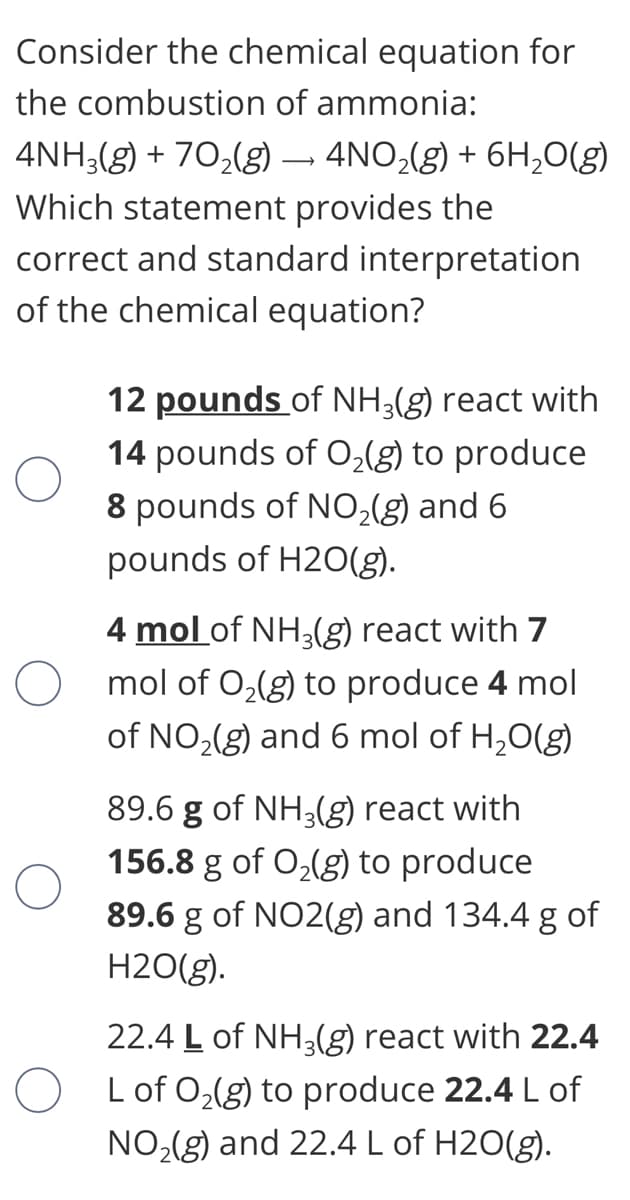
Transcribed Image Text:Consider the chemical equation for
the combustion of ammonia:
4NH3(g) + 70,(g) – 4NO,(g) + 6H,0(g)
Which statement provides the
correct and standard interpretation
of the chemical equation?
12 pounds of NH3(g) react with
14 pounds of O,(g) to produce
8 pounds of NO,(g) and 6
pounds of H2O(g).
4 mol of NH3(g) react with 7
O mol of 0,(g) to produce 4 mol
of NO,(g) and 6 mol of H,0(g)
89.6 g of NH3(g) react with
156.8 g of O,(g) to produce
89.6 g of NO2(g) and 134.4 g of
H2O(g).
22.4 L of NH3(g) react with 22.4
O Lof 0,(g) to produce 22.4 L of
NO,(g) and 22.4 L of H2O(g).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning
