Consider the following reaction occurring at 25C Nitrogen monoxide gas reacts with hydrogen gas to form ammonia gas and water vapor (steam) Use Appendix 2 to obtain-the data required to find the following: (fill in the blanks with a numerical response only. Do not write the units in your answer.) 1. Enthalpy of reaction: AH kJ/mol %3D 2. Entropy of system: AS J/mol 3. Entropy of surroundings: AS J/mol %3D surr 4. Entropy of universe: AS univ J/mol
Consider the following reaction occurring at 25C Nitrogen monoxide gas reacts with hydrogen gas to form ammonia gas and water vapor (steam) Use Appendix 2 to obtain-the data required to find the following: (fill in the blanks with a numerical response only. Do not write the units in your answer.) 1. Enthalpy of reaction: AH kJ/mol %3D 2. Entropy of system: AS J/mol 3. Entropy of surroundings: AS J/mol %3D surr 4. Entropy of universe: AS univ J/mol
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter10: Entropy And The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 10.88PAE: Nickel metal reacts with carbon monoxide to form tetra-carbonyl nickel, Ni(CO)4:...
Related questions
Question
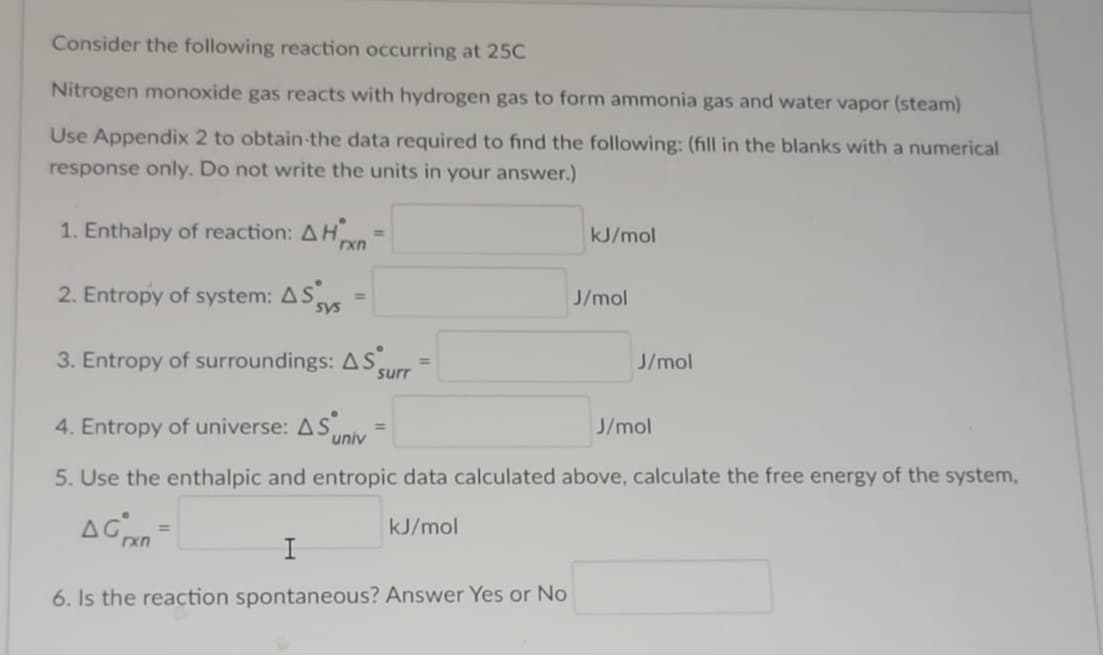
Transcribed Image Text:Consider the following reaction occurring at 25C
Nitrogen monoxide gas reacts with hydrogen gas to form ammonia gas and water vapor (steam)
Use Appendix 2 to obtain-the data required to find the following: (fill in the blanks with a numerical
response only. Do not write the units in your answer.)
1. Enthalpy of reaction: AH
kJ/mol
%3D
2. Entropy of system: AS
J/mol
3. Entropy of surroundings: AS.
J/mol
surr
4. Entropy of universe: ASM
J/mol
%3D
univ
5. Use the enthalpic and entropic data calculated above, calculate the free energy of the system,
AGn
kJ/mol
%3D
I
6. Is the reaction spontaneous? Answer Yes or No
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
