Consider the gas-phase equilibrium A 2B, where both the forward and reverse reactions are elementary processes with rate constants of k and k,, respectively. The concentrations of both A and B were monitored and plotted below as a function of time. B -A time Which of the following statements is/are correct? (select all that apply) ki = k, at equilibrium. ky (A)2 = k, (B] at equilibrium O ky (A) = k, [B}? at equilibrium Kea > 1 for this equilibrium, assuming that both (Aleg and (Bleg are greater than 1.0 M. The rate for the forward reaction A - 2B decreases as the system approach equilibrium. concentration
Consider the gas-phase equilibrium A 2B, where both the forward and reverse reactions are elementary processes with rate constants of k and k,, respectively. The concentrations of both A and B were monitored and plotted below as a function of time. B -A time Which of the following statements is/are correct? (select all that apply) ki = k, at equilibrium. ky (A)2 = k, (B] at equilibrium O ky (A) = k, [B}? at equilibrium Kea > 1 for this equilibrium, assuming that both (Aleg and (Bleg are greater than 1.0 M. The rate for the forward reaction A - 2B decreases as the system approach equilibrium. concentration
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 8ALQ
Related questions
Question
100%
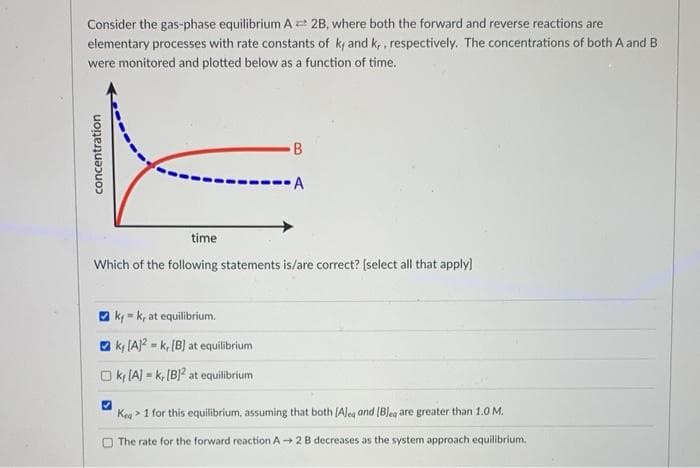
Transcribed Image Text:Consider the gas-phase equilibrium A 2B, where both the forward and reverse reactions are
elementary processes with rate constants of k and k,, respectively. The concentrations of both A and B
were monitored and plotted below as a function of time.
B
-A
time
Which of the following statements is/are correct? (select all that apply)
ky = k, at equilibrium.
O k, (A? - k, (B) at equilibrium
O ky (A) = k, [B}? at equilibrium
Keg > 1 for this equilibrium, assuming that both (Aleg and (Bleg are greater than 1.0 M.
The rate for the forward reaction A + 2 B decreases as the system approach equilibrium.
concentration
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning
