Consider the potential energy diagram shown below. This graph shows the chemical potential energy in a reaction system over time. The y-axis is potential energy in kilojoules. The x-axis is the reaction progress, or time. Does this graph represent an endothermic or an exothermic reaction? Explain your answer. What is the enthalpy change, ΔH, for this reaction? Show your work. What is the activation energy, Ea, for this reaction? Show your work.
Consider the potential energy diagram shown below. This graph shows the chemical potential energy in a reaction system over time. The y-axis is potential energy in kilojoules. The x-axis is the reaction progress, or time. Does this graph represent an endothermic or an exothermic reaction? Explain your answer. What is the enthalpy change, ΔH, for this reaction? Show your work. What is the activation energy, Ea, for this reaction? Show your work.
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter2: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 2.21E
Related questions
Question
It is time to practice using potential energy diagrams. Respond to the three questions below on energy diagrams and submit to your instructor.
- Consider the potential energy diagram shown below. This graph shows the chemical potential energy in a reaction system over time. The y-axis is potential energy in kilojoules. The x-axis is the reaction progress, or time.
- Does this graph represent an endothermic or an exothermic reaction? Explain your answer.
- What is the enthalpy change, ΔH, for this reaction? Show your work.
- What is the activation energy, Ea, for this reaction? Show your work.
- In a particular
chemical reaction , the energy of the reactants is 30 kJ and the energy of the products is 5 kJ. The maximum energy of the system is 40 kJ.- Sketch a potential energy diagram for this reaction. Make sure to label the energy of the reactants, the energy of the products, the activation energy, and the enthalpy change for the reaction.
- What is the activation energy for this reaction?
- What is the enthalpy change for this reaction?
- Is this reaction endothermic or exothermic? Explain your answer in two ways: first, using the energy values, and second, by referring to the shape of the graph.
- The coating on the head of a match is highly flammable. When it burns, it releases a great deal of energy. However, before the match can burn, it must gain a small amount of energy from a spark. That spark is typically produced by striking (rubbing) the match head against a rough surface. Sketch and describe a potential energy diagram that represents the striking and burning of the match. Remember to label the diagram with the energy changes that occur. Your answer must include the potential energy diagram and a written description. (Note: you do not have to use actual energy values.)
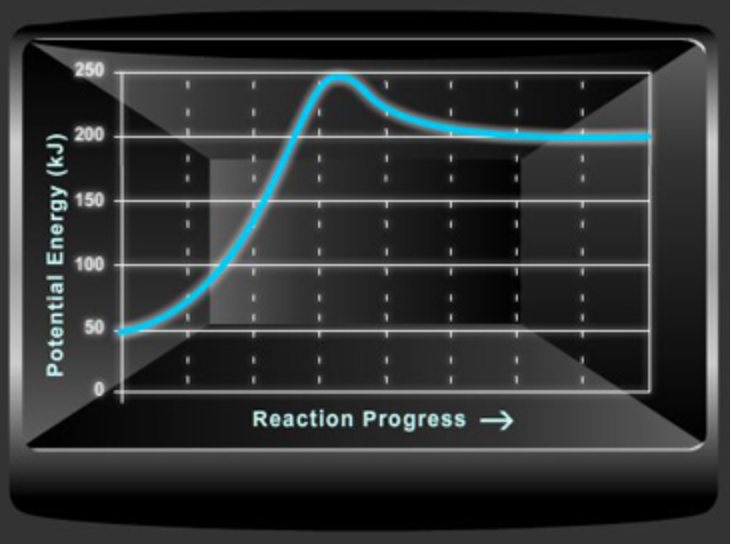
Transcribed Image Text:Potential Energy (kJ)
250
200
150
100
50
Reaction Progress →
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning