Consider the reaction described in the previous problems. Use the following expression, no.1 e-4/2kT %3D n2 to calculate the ratio of H atoms to H2 molecules in a hydrogen gas in thermal equilibrium with a reservoir at 904 K when there are 6 x 1020 molecules m3 of H2. Recall that A = 4 eV and that a convenient approximation for the quantum density is given by no= (1030 m-3)(m/m3/2(T/300 K)3/2, where the value of the temperature in K must be inserted for T, and the ratio of m/m, is the atomic mass (in atomic mass units, amu) for an atom, or the sum of atomic masses (in amu) for atoms in a molecule. Write the numerical answer in exponential form accurate to 2 decimal places, as described in the worksheet instructions (X.XXE+Y or X.XXE-Y).
Consider the reaction described in the previous problems. Use the following expression, no.1 e-4/2kT %3D n2 to calculate the ratio of H atoms to H2 molecules in a hydrogen gas in thermal equilibrium with a reservoir at 904 K when there are 6 x 1020 molecules m3 of H2. Recall that A = 4 eV and that a convenient approximation for the quantum density is given by no= (1030 m-3)(m/m3/2(T/300 K)3/2, where the value of the temperature in K must be inserted for T, and the ratio of m/m, is the atomic mass (in atomic mass units, amu) for an atom, or the sum of atomic masses (in amu) for atoms in a molecule. Write the numerical answer in exponential form accurate to 2 decimal places, as described in the worksheet instructions (X.XXE+Y or X.XXE-Y).
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter5: Principles Of Chemical Reactivity: Energy And Chemical Reactions
Section5.8: Product- Or Reactant-favored Reactions And Thermodynamics
Problem 1.2ACP
Related questions
Question
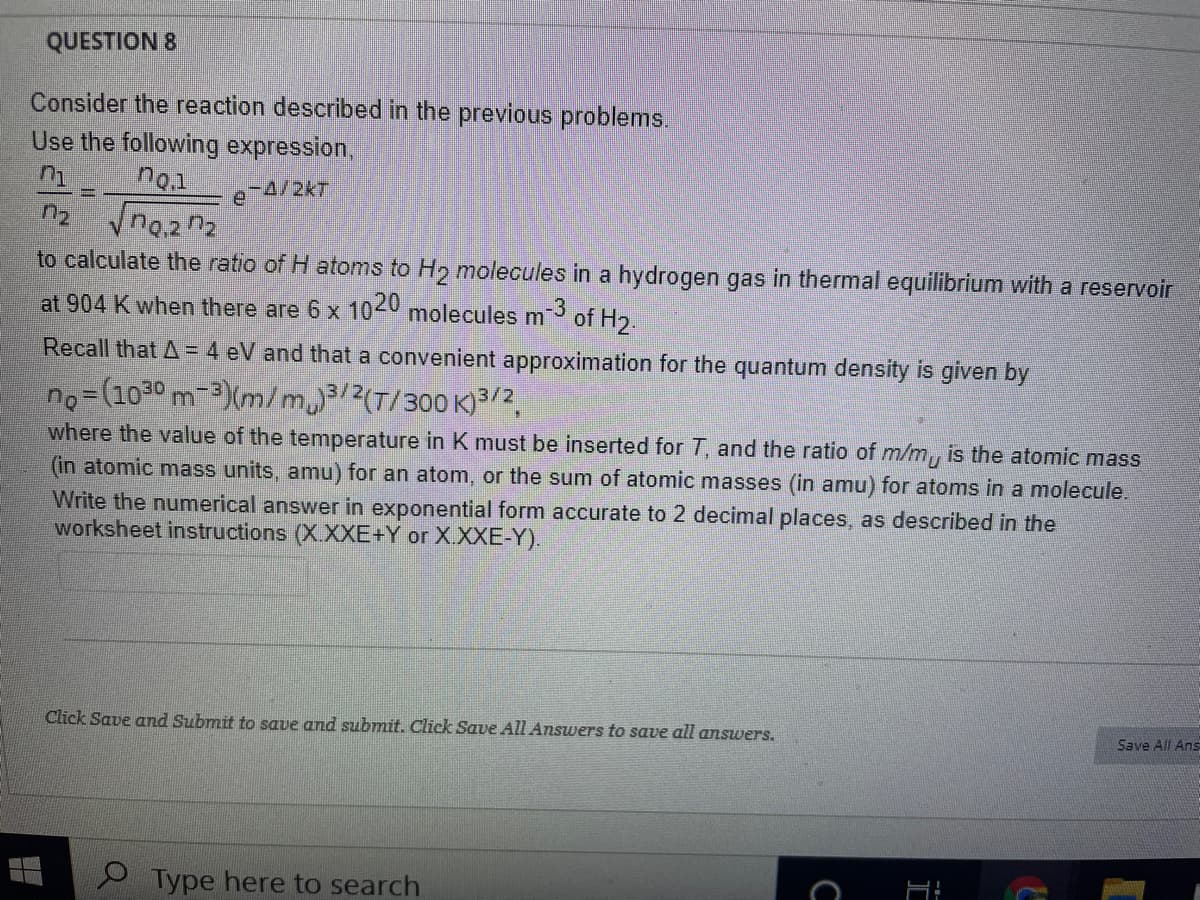
Transcribed Image Text:QUESTION 8
Consider the reaction described in the previous problems.
Use the following expression,
no.1
e 4/2kT
to calculate the ratio of H atoms to H2 molecules in a hydrogen gas in thermal equilibrium with a reservoir
at 904 K when there are 6 x 1040 molecules m
-3
of H2.
Recall that A = 4 eV and that a convenient approximation for the quantum density is given by
no=(1030 m-3)(m/m3/2(T/300 K)3/2,
where the value of the temperature in K must be inserted for T, and the ratio of m/m, is the atomic mass
(in atomic mass units, amu) for an atom, or the sum of atomic masses (in amu) for atoms in a molecule.
Write the numerical answer in exponential form accurate to 2 decimal places, as described in the
worksheet instructions (X.XXE+Y or X.XXE-Y).
Click Save and Submit to save and submit. Click Save All Answers to save all answers.
Save All Ans
Type here to search
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning