Consider the reaction of 1.41 g Ca(s) with 107 mL of 0.325 M HBr(aq) according to the following unbalanced chemical equation: Ca(s) + HBr(aq) → CaBr(aq) + H2(g) The hydrogen produced was collected by displacement of water at 22°C with a total pressure of 759 torr. Which reactant is the limiting reagent? HBr O Ca Correct Balanced chemical equation: Сa() + 2HBr(aq) — СаBrz(aq) + H>(e) HBr is the limiting reactant. Ca(s) + HBr(aq) – CaBr2(aq) + H2(g) Determine the volume (in L) of hydrogen gas produced if the vapor pressure of water at 22°C is 21 torr. L How many grams of the other reactant are left over?
Consider the reaction of 1.41 g Ca(s) with 107 mL of 0.325 M HBr(aq) according to the following unbalanced chemical equation: Ca(s) + HBr(aq) → CaBr(aq) + H2(g) The hydrogen produced was collected by displacement of water at 22°C with a total pressure of 759 torr. Which reactant is the limiting reagent? HBr O Ca Correct Balanced chemical equation: Сa() + 2HBr(aq) — СаBrz(aq) + H>(e) HBr is the limiting reactant. Ca(s) + HBr(aq) – CaBr2(aq) + H2(g) Determine the volume (in L) of hydrogen gas produced if the vapor pressure of water at 22°C is 21 torr. L How many grams of the other reactant are left over?
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter5: Gases
Section: Chapter Questions
Problem 5.93PAE: 93 The complete combustion of octane can be used as a model for the burning of gasoline:...
Related questions
Question
100%
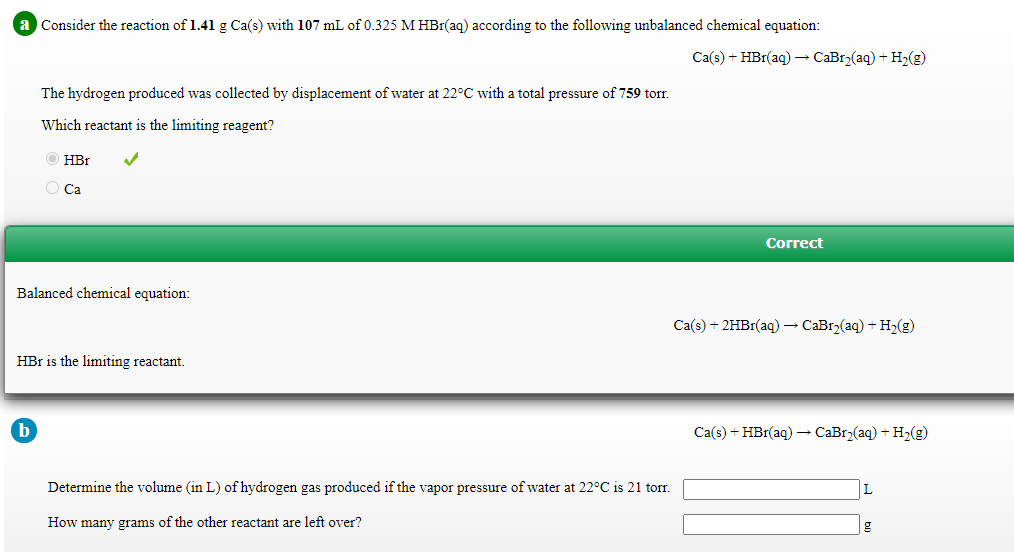
Transcribed Image Text:a Consider the reaction of 1.41 g Ca(s) with 107 mL of 0.325 M HBr(aq) according to the following unbalanced chemical equation:
Ca(s) + HBr(aq) → CaBr2(aq) + H2(g)
The hydrogen produced was collected by displacement of water at 22°C with a total pressure of 759 tor.
Which reactant is the limiting reagent?
HBr
O Ca
Correct
Balanced chemical equation:
Ca(s) + 2HB1(aq) – CaBr2(aq) + H2(g)
HBr is the limiting reactant.
Ca(s) + HBr(aq) → CaBr2(aq) + H2(g)
Determine the volume (in L) of hydrogen gas produced if the vapor pressure of water at 22°C is 21 torr.
L
How many grams of the other reactant are left over?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning