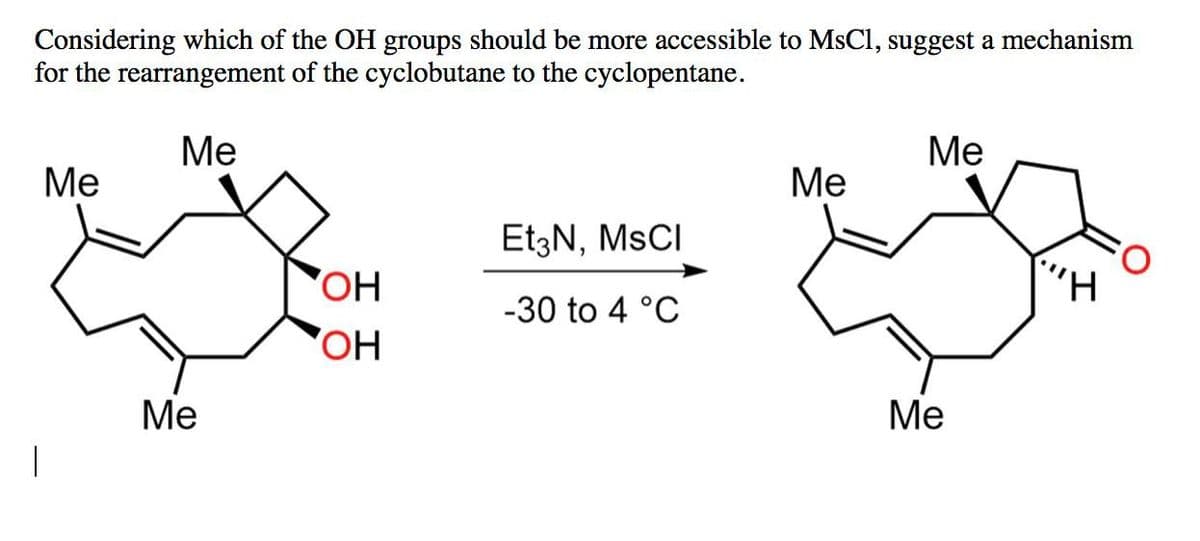
Considering which of the OH groups should be more accessible to MsCl, suggest a mechanism for the rearrangement of the cyclobutane to the cyclopentane. Me Me Me Me Et3N, MSCI OH -30 to 4 °C OH Me Me
Q: Question 25 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: If you find this answer helpful please rate it good. If any query then raise it.Thank You!
Q: solve step by step
A: Detailed explanation:Part (a): Composition of the Liquid MixtureWe are given:Vapor pressure of…
Q: In order to diagnose the congenital chromosomal disorder Turner's syndrome, clinical symptoms (neck…
A: Title: Diagnostic Criteria and Hormonal Profiles in Turner Syndrome: A Comprehensive…
Q: 11. What substitution reaction mechanism is most likely for the following compound? Br A) B) SN1 SN2…
A:
Q: d. H3N H₂C!!!! H3CW H H-N 'N' Ph 4|1!!! HCl(aq)
A: Step 1: Identify the Central Atom and ChargeThe central atom is Nitrogen (N) with three single…
Q: from the list, match a molecule (only one for each case) to the IR spectrum A and B.
A: Spectrum AI only encircle the necessary peaks to identify the molecule. #1 indicate a peak for O-H…
Q: None
A: Step 1:(a)Let me explain the net ionic equation for the reaction between chromium(III) chloride and…
Q: Question 12 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: In a negative feedback system like thyroid hormones, when the target hormones (T3, T4) are…
A: The statement is FALSE. Let's take a look at these diagrams of the Thyroid Hormone Feedback Loop…
Q: CIO (aq) + Cr(OH)3(s) CrO42(aq) + Cl¯(aq) basic What is the standard cell potential, E° cell, for…
A: Step 1:The standard electrode potential, often referred to as standard potential, indicates the…
Q: How would you answer part III of this FRQ? this is a non graded practice.
A: (iii) Polar and Non-Polar Molecules: Iodine Monofluoride (IF₅):Iodine monofluoride has a trigonal…
Q: Question 27 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: None
A: The two compounds in the image are constitutional (structural) isomers. So the answer is option…
Q: Cl H H I -H Identify the 'cartoon' drawing of the donor orbital in the nucleophilic substitution…
A: The text describes a reaction between ethanoyl chloride (CH3COCl) and lithium aluminum hydride…
Q: 3. Binary system acetonitrile(1)/nitromethane(2) conforms closely to Raoult's law. Vapor pressures…
A: Sure, I can help you with this problem which asks you to prepare a Pxy diagram for a binary system…
Q: Please classify and explain the following as either aromatic (A) or nonaromatic (NA)
A: Step 1:To answer this question, we must be familiar with the 4 laws of aromaticity. 1. It must be…
Q: For the random two substrate binding reaction shown here: ΚΑ R+A RA + B + B KB 11 RB + A ΚΑ Jak KB…
A:
Q: Draw the structure(s) of the major organic product(s), including counterions, of the following…
A:
Q: Predict the missing product of this organic reaction please:
A: Step 1: Step 2:Mechanism Step 3: Step 4:
Q: None
A: a. 1,2-dihydroxybenzene (common name: catechol) - This compound is a benzene ring with two hydroxyl…
Q: Outline the steps you would take to conduct a thorough risk assessment of chemical hazards in the…
A: Analyzing the approach to conducting a thorough risk assessment of chemical hazards in the workplace…
Q: (a) A voltaic cell is constructed as follows: Zn | Zn2+ | H+ | H2 The half-cell reactions are:…
A: i. Given: Ecell°=???V;Ecell=0.45V;[Zn2+]=1.0M;PH2=1.0atm;[H+]=???MZn(s)∣Zn2+∣∣H+∣H2(g)Step 1:…
Q: Draw the structure of the major organic product of the reaction. CI (CH3)2₂CuLi ether, -78° C Marvin…
A: Gilman's reagent, typically composed of an organocopper compound such as lithium organocopper…
Q: Determine whether the following are aromatic, nonaromatic or antiaromatic.
A: Thank you.
Q: 1) Which of the following resonance structures of DMAP are wrong? N N DMAP A N B N + N N= N + C D
A:
Q: Question 26 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: To predict the final product of a multi-step organic synthesis reaction, let's go through the…
Q: In a constant-pressure calorimeter, 60.0 mL of 0.810 M H2SO4 was added to 60.0 mL of 0.370 M NaOH.…
A: For No. 1: The total volume of the mixture assuming the total volume is the sum of the individual…
Q: None
A: A Diels-Alder reaction is a [cycloaddition] reaction in organic chemistry that occurs between a…
Q: Draw a structural formula for the alkene you would use to prepare the alcohol shown by…
A: Step 1: Step 2: Mechanism Step 3:Step 1: HydroborationThis step involves the reaction of the alkene…
Q: What is the systematic name for the molecule below? O 2,3-dimethyl-1-ethylcyclohexane O…
A: So, the systematic name for the given molecule is: 1-ethyl-2,3-dimethylcyclohexane
Q: Please answer the following chem question
A:
Q: Complete solutions need all solve
A: The preparation of standard solutions through the direct method involves calculating the required…
Q: For each of the following antibiotics, determine how many moles of each compound are present for the…
A: The objective of this question is to calculate the number of moles of tetracycline (C22H24N2O8)…
Q: Write the systematic name of each organic molecule: structure CH3 CI- CH-CH2- CH=CH2 CH2 CI C-CH2- -…
A: Thank you.
Q: How much energy must be supplied to break a single aluminum-27 nucleus into separated protons and…
A: The objective of this question is to calculate the energy required to break a single aluminum-27…
Q: None
A:
Q: Draw the major organic product(s) of the following reactions including stereochemistry when it is…
A: Step 1:In the presence of H₂SO₄/HgSO₄, the alkene (CH₃CH₂C=CCH₃) undergoes protonation. The acidic…
Q: What is the enegry numbers for the table?1 calorie (cal) = 4.184 J1 kWh = 3.600 × 106 J
A: Please see the attached image for the solution. If there are queries,please do not hesitate to ask.…
Q: None
A: Answer: O-C≡OClF5Hybridisationspsp3d2Electronic geometryLinearOctahedralMolecular…
Q: 4. Show the mechanism for the following transformation. As always, be sure to show all intermediates…
A: References Smith, M. B. (2020). March's advanced organic chemistry: reactions, mechanisms, and…
Q: Please don't provide handwritten solution ....
A: Approach to solving the question: - Detailed explanation: Given above Examples: - Key references: -
Q: 45) Determine whether each pair of substances forms a homogeneous solution when combined. For those…
A: A) LiF and C6H14 (Lithium Fluoride and Hexane)LiF is an ionic compound (metal and non-metal).C6H14…
Q: Determine the predominant type of reaction: SN1, SN2, E1 and E2. Draw the major products only.…
A:
Q: Question 7 Propose a chemical structure for the name below. Make sure to clearly define the…
A:
Q: Identify the circled functional group(s) for each molecule. OH CH3CHCH3 a. ΙΟ ——CH2 b.…
A: functional group is an atom, or a group of atoms present in a molecule that is responsible for the…
Q: Can you write down retrosynthesis ( RSA)and show pilar dont provide hnadwriting solution...
A: Step 1: Bromobenzene's Retrosynthesis : * Target Molecule : C6H5Br (bromobenzene). * Functional…
Q: In the following FTIR Spectrum, what functional group can you recognize?
A: Step 1: Step 2: Step 3:
Q: Question 5 Which set of Newman projections represents correct the most stable and the least stable…
A: Question 5: The most stable is when they CH3 are anti to each other180∘. Whereas the least stable…
Q: None
A: Step 1:Find total valence electrons : Total number of valence electrons in SO3= 6 + (3×6)= 24 Step…
Q: Draw the starting alkyne that would lead to the following two products under these conditions.…
A:
Step by step
Solved in 2 steps with 1 images

- Calculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4A solution of pure (S)-2-iodobutane ([a] = +15.90°) in acetone is allowed to react with radioactive iodide, 131I -, until 1.0% of the iodobutane contains radioactive iodine. The specific rotation of this recovered iodobutane is found to be +15.58°. (a) What does this result suggest about the mechanism of the reaction of 2-iodobutane with iodide ion?8 Give logical fragmentation reactions to account for the following ions observed in these mass spectra.(a) n-octane: 114, 85, 71, 57 (b) methylcyclohexane: 98, 83 (c) 2-methylpent-2-ene: 84, 69(d) pentan-1-ol: 70, 55, 41, 31
- Qa 18. Bromination of 5a-cholestan-3-one occurs at C-2 or C-4 to give 2 regioisomers. Two unsaturated ketones with lambdamax = 230 nm and lambdamax = 241 nm are yielded during dehydrobromination. Use Woodward-Fieser rules to distinguish between the two.at 25C, which of the following reactions of PbS and H2O2 is more favourable?1) PbS + 4H2O2 -> SO2 + PbO2 + 4H2O2) PbS + H2O2 -> PbSO4 + H2OVirgin coconut oil contains about 46-48% of lauric acid (CH3(CH2)10COOH). In thepresence acid catalyst and an alcohol, propose the mechanism of this reaction.
- The reaction of 2,2-dimethyl-1-propanol [(CH3)3CCH2OH], also known by the common name neopentyl alcohol, with HBr is very slow and gives 2-bromo-2-methylbutane as the major product.Give a mechanistic explanation for these observations.Cembrene, C20H32, is a diterpenoid hydrocarbon isolated from pine resin. Cembrene has a UV absorption at 245 nm, but dihydrocembrene (C20H34), the product of hydrogenation with 1 equivalent of H2, has no UV absorption. On exhaustive hydrogenation, 4 equivalents of H2 react, and octahydrocembrene, C20H40, is produced. On ozonolysis of cembrene, followed by treatment of the ozonide with zinc, four carbonylcontaining products are obtained: Propose a structure for cembrene that is consistent with its formation from geranylgeranyl diphosphate.Conformation control of reaction mechansims. Consider the reaction below: Provide all possible elimination products from this reaction and identify the major elimination product. Provide mechanisms (with curved arrows) that clearly explain the reaction outcome.
- I need help proposing a detailed electron pushing mechanism showing the cyclinization of the amide intermediate to benzimidazole. I must show all resonance stabilized carbocations with RSCC letters. Please help!Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answerGive logical fragmentation reactions to account for the following ions observed in these mass spectra. (a) 2-methylpent-2-ene: 84, 69 (b) pentan-1-ol: 70, 55, 41, 31

