Constants It takes 53.0 J to raise the temperature of an 8.40 g piece of unknown metal from 13.0°C to 25.0°C What is the specific heat for the metal? Express your answer with the appropriate units. View Available Hint(s) C₂0 526 J-(g-°C)¹ Previous Answers Correct Parts B and C The next two questions pertain to silver. They have nothing to do with unknown metal described in Part A Part B The molar heat capacity of silver is 25.35 J/mol C. How much energy would it take to raise the temperature of 8 40 g of silver by 13.9°C Express your answer with the appropriate units. > View Available Hint(s) MA ? 9 =
Constants It takes 53.0 J to raise the temperature of an 8.40 g piece of unknown metal from 13.0°C to 25.0°C What is the specific heat for the metal? Express your answer with the appropriate units. View Available Hint(s) C₂0 526 J-(g-°C)¹ Previous Answers Correct Parts B and C The next two questions pertain to silver. They have nothing to do with unknown metal described in Part A Part B The molar heat capacity of silver is 25.35 J/mol C. How much energy would it take to raise the temperature of 8 40 g of silver by 13.9°C Express your answer with the appropriate units. > View Available Hint(s) MA ? 9 =
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter12: Thermodynamic Processes And Thermochemistry
Section: Chapter Questions
Problem 16P
Related questions
Question
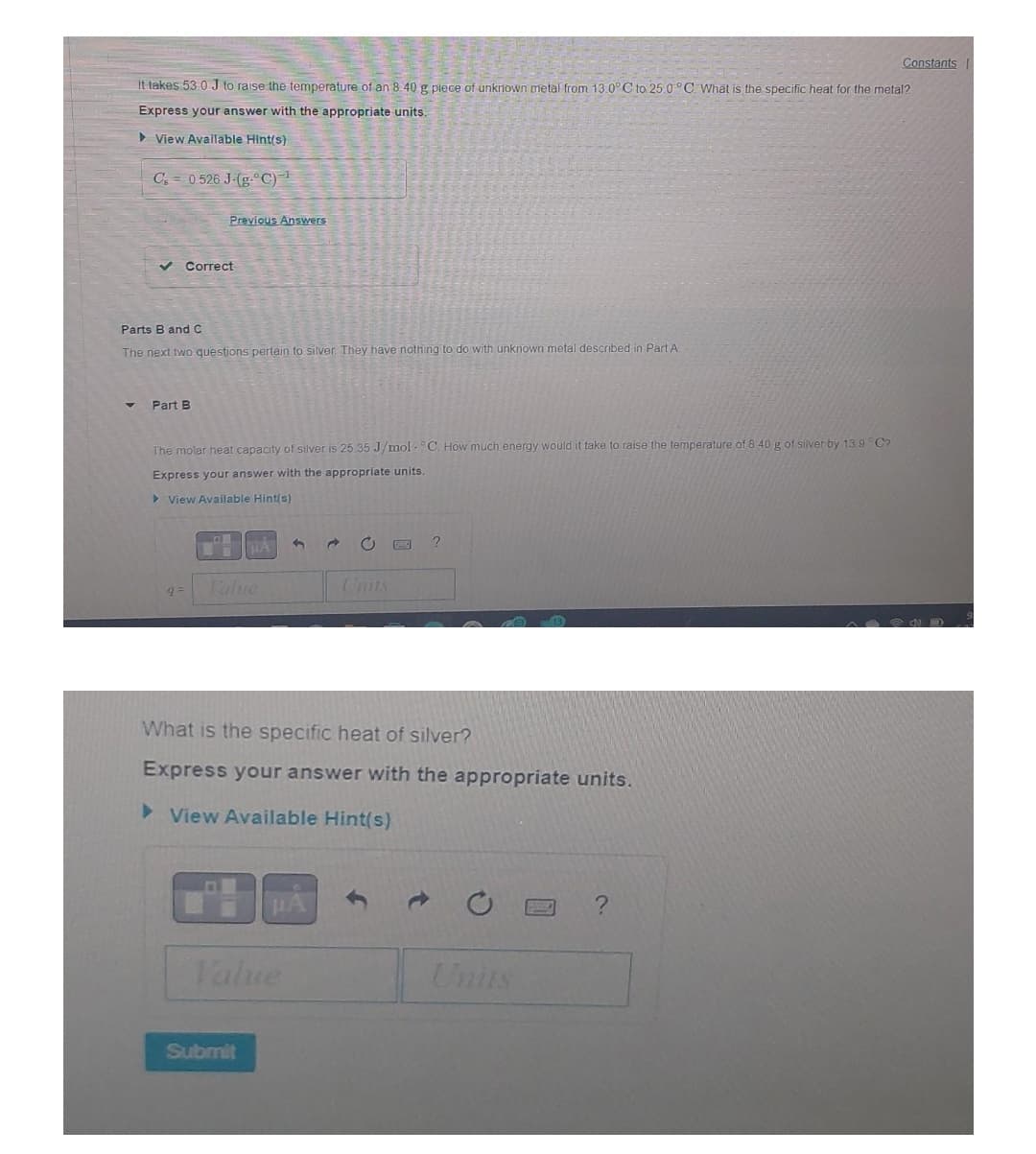
Transcribed Image Text:Constants
It takes 53.0 J to raise the temperature of an 8.40 g piece of unknown metal from 13.0°C to 25.0°C. What is the specific heat for the metal?
Express your answer with the appropriate units.
View Available Hint(s)
C 0 526 J-(g-°C)¹
Previous Answers
✓ Correct
Parts B and C
The next two questions pertain to silver. They have nothing to do with unknown metal described in Part A
Part B
The molar heat capacity of silver is 25.35 J/mol °C. How much energy would it take to raise the temperature of 8 40 g of silver by 13.9°C
Express your answer with the appropriate units.
View Available Hint(s)
μA
C
?
9 =
Value
What is the specific heat of silver?
Express your answer with the appropriate units.
► View Available Hint(s)
μÅ
?
Value
Submit
3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning