Course Contents»... LA G Liquid hellum at 4.2 K has a density of 0.147 g/mL. Suppose that a 1.00 L metal bottle at 80 K contains air at 2.0 atm pressure. If we introduce 150.0 mL of liquid hellum, seal the bottle, and allow the entire system to warm to room temperature (25°C), what is the pressure inside the bottle (in atmospheres)? Submit Answer Tries 0/99
Course Contents»... LA G Liquid hellum at 4.2 K has a density of 0.147 g/mL. Suppose that a 1.00 L metal bottle at 80 K contains air at 2.0 atm pressure. If we introduce 150.0 mL of liquid hellum, seal the bottle, and allow the entire system to warm to room temperature (25°C), what is the pressure inside the bottle (in atmospheres)? Submit Answer Tries 0/99
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter4: Introduction To Gases
Section: Chapter Questions
Problem 33E
Related questions
Question
100%
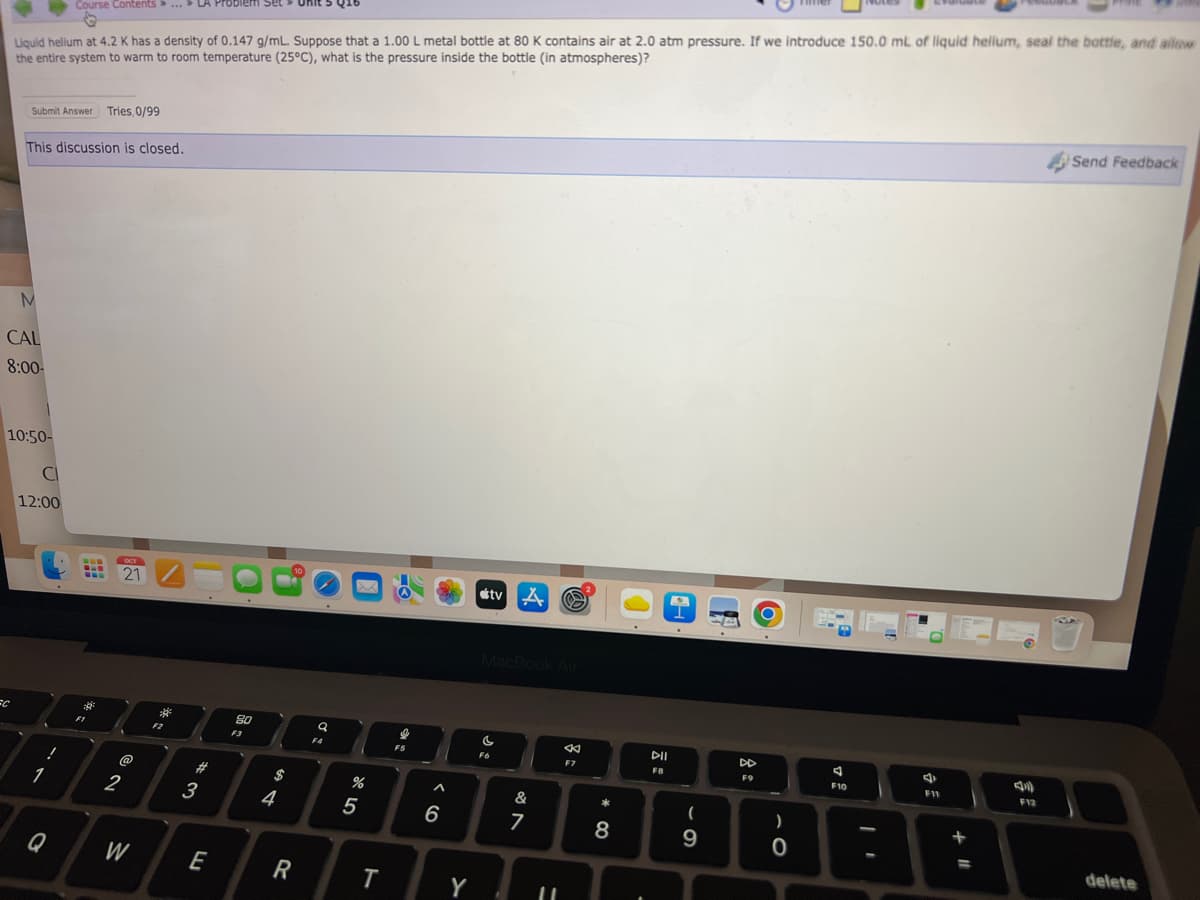
Transcribed Image Text:Liquid helium at 4.2 K has a density of 0.147 g/mL. Suppose that a 1.00 L metal bottle at 80 K contains air at 2.0 atm pressure. If we introduce 150.0 mL of liquid helium, seal the bottle, and allow
the entire system to warm to room temperature (25°C), what is the pressure inside the bottle (in atmospheres)?
=C
Submit Answer Tries, 0/99
M
CAL
8:00-
This discussion is closed.
10:50-
CI
12:00
Course Contents»... LA
G
1
!
Q
. BUD
B
R
F1
ост
21
@
2
W
3
F2
*3
#
E
80
F3
$
4
10
R
Q
F4
%
5
T
FS
A
6
Y
tv A a
MacBook Air
F6
&
7
=
8
F7
*
8
DII
FB
(
9
=
DD
F9
)
0
A
F10
F11
+
=
F12
Send Feedback
4
delete
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning