Q: Sketch energy diagram for a one-step reaction that is fast and highly exergonic.
A: Those reactions in which energy is released are called exergonic reactions .
Q: 13. (a) Compound C undergoes a reaction with SOCI2 (or PCI5) to yield compound D (Molecular Formula…
A: This Question is based upon NMR Spectroscopy.
Q: Indicators are acids or bases whose various protonated forms have different colors. The volume…
A: Acid is a substance that give hydrogen ions in aqueous solution and base is a substance that give…
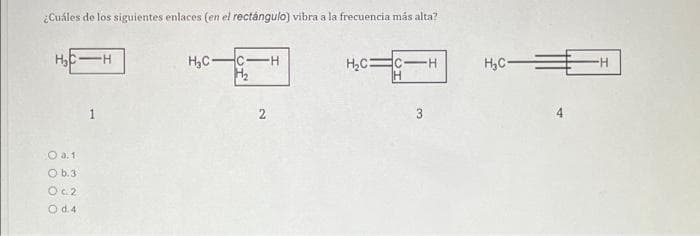
Q: Los siguientes datos fueron obtenidos para la reacción hipotética: 2A + B - productos Rapidez…
A:
Q: The Ecell of a reaction were found to be +0.78 V. What is the spontaneity of the reaction. none of…
A:
Q: Arrange the valence molecular orbitals of AB in decreasing energy (1 highest in energy; 6 lowest in…
A: Consider a Heteronuclear diatomic Molecule ,AB where A and B are period of 2 elements According to…
Q: These two structures, shown as Fischer projections, are: Br H3C- H- H-N- H- NH2 Br the same compound…
A: Stereisomers are those which have same molecular formula but different arrangement of atoms in…
Q: onsider the reaction of acetic acid (HC2H3O2) in vinegar with NaOH solution 1) write a balanced…
A: Here we are required to find the mass % of acetic acid in vinegar.
Q: A gas will behave ideally as long as it is at a high and a low A) temperature; pressure B) pressure;…
A:
Q: 2. Calculate reaction AH°rxn and reaction AU°rxn for the total oxidation of benzene. Use the stable…
A: Here we have to determine ∆H and ∆U for the complete combustion of benzene at 298.15K.
Q: Chemical properties of materials include
A:
Q: true or false A desirable solvent should be highly pure, commercially available, low viscosity and…
A: The given statement is " A desirable solvent should be highly pure, commercially available, low…
Q: A solution of HA (0.277M; 64.1mL; K, =5.5E-5) is titrated with 0.8033M KOH. Determine the pH at the…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Chemistry draw the structures of soap and identify its components.
A: Soap molecules consists of following two parts.
Q: A gas is under pressure of pressure 20.855 bar gage, T= 104 Fahrenheit and unit weight is 362 N/m3.…
A: Given that , P= 20.855 bar T = 104 oF Unit weight = 362 N/m3
Q: 4. Draw an energy level diagram for bromine
A: Energy level diagram for bromine
Q: A gas will behave ideally as long as it is at a high and a low A) temperature; pressure B) pressure;…
A:
Q: a. The nitroprusside test is a test for the detection of methyl ketones. What are the reagents used…
A:
Q: Q3/If we have more than one substance with a large difference in melting point and we want to…
A:
Q: ulate the value ol KC Ior ne reaction: PC|3 (g) Cl2 (g) AH = Positive PCI5 (g) + Given that when 8.4…
A:
Q: Calculate the absorbance of an organic dye [C =7×10-4 mol L−1] ,knowing that the molar absorptivity…
A: Given: The molar concentration of the solution = 7×10-4 mol L−1 The molar absorptivity, ∈= 650 mol…
Q: What characteristic charge do atoms in the third-to-last column of the periodic table have when they…
A: We will explain this statement with the help of morden periodic table.
Q: 3. Calculate reaction AH°rxn and reaction AU°rxn for the reaction: TiCl4(1) + 2H20(1) → TIO2(s) +…
A:
Q: Which type of spectroscopy would be best/easiest to distinguish between these two compounds? CH3 CH3
A: Different spectroscopic techniques are there which helps us to understand a unknown organic…
Q: Which of these would give a positive iodoform (I2, NaOH) test? CH3 CH3 H;C H;C H3C H. H3C C A OB O…
A:
Q: The redox titration reached the endpoint when 0.01924 mol triiodide solution were added. 15(aq) +…
A:
Q: CH3 Write in the correct reagents for these transformations: a) cyclohexanol chlorocyclohexane b)…
A: Here we are required to predict the reagent needed to carry out the following conversion
Q: The hybridization (sp? sp??sp3? sp4?) of the carbon in formic acid is and the bond angles around the…
A: Steric number = no. of bond pair + no. of loan pair ->steric number = 2=> sp =3 =>…
Q: Provide the structure of the product(s) with stereochemisty if appropriate. If multiple products…
A:
Q: 1ll 111 Sose the answers from the box. energy work internal energY increased sum 1. The first law of…
A: Solution: 1) The first law of thermodynamics applies the conservation of ENERGY…
Q: a.) Define what is a salt bridge b.) what is the role of the salt bridge in galvanic cells? c.)…
A: Salt brige is used in galvanic cell, it has a great role in electrochemistry.
Q: Check Your Understanding Fill in the blanks in the paragraph below. In Fig. 7-2, if the piston was…
A: Given:
Q: The name of the starting compound that gives these compounds upon treatment with ozone, followed by…
A:
Q: Consider the compounds belowz show how Hinsberg's Method for Characterizing Primary, Secondary, and…
A: Hinsberg test is used to distinguish primary, secondary and tertiary amine.
Q: 2-Assign R or S configuration to the chirality center in each of the following molecules: H. H-CIOH…
A: R & S configurations are for enantiomers. Only even changes are allowed.
Q: 2. The rate constant for the first-order reaction: SO2Cl2(g) = SO2(g) + Cl2(g) is 2.2 x 10–5 s --1.…
A:
Q: . Given that the crystal structure of Manganese iodide. It showed 21 bonds at 452 pm and 41 bonds at…
A: The shape of transition metal complexes is determined by the tendency of electron pairs to occupy…
Q: Write in the correct reagents for these transformations: a) cyclohexanol - -→ chlorocyclohexane b)…
A:
Q: 1. Melting point technique 2. Recrystallization technique| 3. Extraction (understand how to separate…
A: As per the guideline, since you have asked multiple questions, we have solved the first question for…
Q: Consider the disulfide bond formation reaction via the oxidation of thiols along with the H-NMR…
A: The proton NMR helps to find the no of different types of protons present in a given molecule.
Q: Differentiate direct dyeing from ingrain dyeing. Considering the structure of cotton and Sudan-1,…
A: Dye is a chemical compound which gives colour. It is used for the coloration of the papers, fabric…
Q: 1. Draw the best Lewis dot structure for SeF4 in the correct molecular geometry [Include formal…
A: SeF4 is an inorganic compound which has 4 highly electronegative F atoms and 1 least electronegative…
Q: . The ammonium ion (NH4+) reacts with nitrite ion (NO2-) to yield nitrogen gas and liquid water. The…
A: Ammonium ion and nitrite ion reacts to produce nitrogen gas and water.
Q: Catculate the EMS stondanet state at 298k for this reaction +2 > Fe + Ti The Units Cam be n v, /,mvJ…
A: Given reaction is : Fe (s) + Ti3+ ---------> Fe2+ + Ti+ Find EMF of the cell at 298 K = ?
Q: (b) 1. SOCI2 H. OEt ОН 2. NaN3, heat 3. ELOH i) draw the products after the first and second step…
A: The first step is the conversion of carboxylic acid to acyl chloride by reacting with thionyl…
Q: Find the pH of a 0.0025 M HCl solution.
A:
Q: H H CH3 CH3 and H TH. H CH3 CH3 H These two compounds can be labeled as: stereoisomers Ob.…
A: The compounds with the same molecular formula but different bond connectivity are structural…
Q: A 25.00 mL aliquot of 0.100 M NH3 is titrated with 21.00 mL of 0.0900 M HCI. Calculate the pH. Kb =…
A: In this question, we will determine the pH of the Buffer solution which is make by after mixing of…
Q: Name the following compounds: CH2 = CH-CH2-0-CH2-C H= CHe %3D
A:
Q: 2. Calculate reaction AH°rxn and reaction AU°rxn for the total oxidation of benzene. Use the stable…
A: Here we have to find ∆H°rxn and ∆U°rxn of combustion of benzene at 298.15K.
Step by step
Solved in 2 steps with 2 images

- Calculate ΔSsys° (J/K) for the catalytic hydrogenation of acetylene to ethane: C2H2(g) + H2(g) → C2H4(g) Substance S° (J/K·mol) C2H2(g) 200.8 H2(g) 130.58 C2H4(g) 219.4In his classic studies of stereochemistry and optical activity in organic compounds, Pasteur measured the optical activity of many solutions. For the naturally occurring enantiomer of tartaric acid, [α]D20 = +12.4o (c = 20, H2O). What can be concluded about the ratio of tartaric acid enantiomers present in the solution if the observed rotation is (i) α = -6.0o, or (ii) α = 0o?In his classic studies of stereochemistry and optical activity in organic compounds, Pasteur measured the optical activity of many solutions. For the naturally occurring enantiomer of tartaric acid, [α]D20 = +12.4o (c = 20, H2O). What can be concluded about the ratio of tartaric acid enantiomers present in the solution if the observed rotation α = 0o?
- Polypeptides are polymers of amino acids. Suppose that a long polypeptide chain can undergo a transition from a helical conformation to a random coil. Consider a mechanism for a helix–coil transition that begins in the middle of the chain:hhhh…⇌ hchh… hchh… ⇌ cccc… in which h and c label, respectively, an amino acid in a helical or coil part of the chain. The first conversion from h to c, also called a nucleation step, is relatively slow, so neither step may be rate determining. (a) Set up the rate equations for this mechanism. (b) Apply the steady-state approximation and show that, under these circumstances, the mechanism is equivalent to hhhh… ⇌ cccc….I am not sure how they got 10 ns^-1, this is occurring at SATP conditions. .subunit G.H.I please
- Predict the signs of rHo and rSo for the transformation: CH3OH(l) ->CH3OH(g) A.) rHo > 0 and rSo > 0 B) rHo < 0 and rSo > 0 C) rHo > 0 and rSo < 0 D) rHo < 0 and rSo < 0For the reaction 2Na (g) Na2 (g) the following values of K have been determined (C. T. Ewinget al., J. Chem. Phys. 1967, 71, 473):T (K) K900 1.321000 0.471100 0.211200 0.10From these data, estimate rxnH° for the reaction.What is known as Ziegler–Natta catalysts ?



