Q: Figure 11 A в E D
A: Where is the correct point to start numbering the structure in Figure
Q: the W the att Vactive between two 12 13 adjacent ions of an atom is 14 15 Jand the rePuisive energy ...
A: Attraction is the stabilization energy and repulsion is disstabilization energy. Binding energy is ...
Q: For each of the following Levey-Jennings charts, circle the choice “acceptable” or “out of control”....
A: Levey-Jennings Control Chart 3 and 4 will go for Acceptable. It is within the Limits Of Standard d...
Q: O KINETICS AND EQUILIBRIUM Deducing a rate law from the change in concentration over time OD O OO Br...
A: If you see the reaction the reaction is called dissociation reaction because Cl2O5 dissociated to pr...
Q: 5. Which one is the following molecules has tetrahedral carbon atom? он H,N LOH B) D) OA
A: We have to identify the one molecule from the following that which molecule have tetrahedral carbon...
Q: Select the single best answer. Decide which conformation is present in higher concentration at equil...
A:
Q: Convert the following measurement. mol 8 6.43 x 10 mol kg m g•cm ||
A:
Q: CH3B + NaOH CH3OH+NaE CH3CH2BR→H2C=CH2 + HBr H2C=CH2 + HCICH3CH2CI Addition Elimination Substitution...
A:
Q: A solution contains 3.85 g of a nonvolatile pure hydrocarbon in 95 g acetone. The boiling points of ...
A: We have to calculate the molar mass of hydrocarbon.
Q: A student added 1.78g of Ca(OH)2(9) to 0.250 L of 0.200 molL"' HNO3(aq)- The mixture was carefully s...
A: We have to calculate the pH of solution.
Q: OH H2 C CH2 H C CH2 H2C CH2 H2C H2 H2
A:
Q: What is the electron configuration of a magnesium atom? 1s22s²2p°2d² O 2s 2s²6p²2s³ O 1s22s²3s²2p O ...
A: The atom given is magnesium.
Q: CH;OH A O HO SH;OH но он OH O CH,OH он CH;OH OH OH CH,OH CH2 CH,OH CH2OH но О Он CH;OH OH OH OH Он O...
A: Amylose and amylopectin are two different forms of starch. Amylose is composed of unbranched chains ...
Q: 1. write out the mechanism for the reaction between ethyne and NaNH2. Label the acid snd the base pr...
A: 1) The reactants given are ethyne and NaNH2.
Q: The following is generic depiction of a reaction using the curve arrow formalism. -D Which of these ...
A:
Q: 1. Based on the general IUPAC rules, which one of the following structures is 4-hydroxybutanal? Но. ...
A: Given : Different structures of the compounds are given. To find : correct structure of 4-hydroxybu...
Q: Draw the structures (condensed) of the following compounds: a. 2-chloro-3,3dimethylhexane b. 3-bromo...
A: Condensed formula of organic compound: Condensed formula of the organic compound can be written...
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D b...
A:
Q: convert 248 mg of Pt to mol of Pt.
A:
Q: Methanol (CH3OH) is manufactured industrially by the reaction: CO(g) + 2H2(g) - CH3OH(g) The equilib...
A: The equilibrium reaction taking place is given as, => CO (g) + 2 H2 (g) -------> CH3OH (g) Gi...
Q: Explain the following terms with a suitable example and/or diagram: (i) Steric and torsional strains...
A: Steric Strain is resulting from the fact that bulky substituents occupy large space around a carbon ...
Q: A pharmacists measures out three amount of 200 mL and two amount of 150 mL from a stock bottle of 2 ...
A: Given : Total volume of stock solution = 2 L. Total mass of a drug = 5 gram...
Q: Barium sulfate is formed from reacting barium hydroxide and sulfuric acid. Indicate the ion/s that a...
A:
Q: A hollow square metal tube has a measurement of 2.42 in for each side and a weight of 53.71 g. The v...
A:
Q: Which of the following outcomes would be most likely if the Hershey-Chase experiments were repeated ...
A: Hershey-Chase were two scientists and their experiment defines DNA as the genetic material rather th...
Q: A solution contains 30 wt% water, 25 wt% ethanol, 15 wt% methanol, 12 wt% glycerol, 10 wt% acetic ac...
A: Given data is as follows: The mass of water = 30 g The mass of ethanol = 25 g The mass of methanol =...
Q: is this polar or non polar?
A:
Q: Explain how ionic bonding, hydrogenbonding, and ion-dipole forced contribute to solutions in enthalp...
A: Heat of hydration: It is the heat that is released by hydration of one mole of ions at a constant p...
Q: You are given the following Born-Haber cycle information. Based on the values listed, determine the...
A:
Q: Suppose you measure a volume of 9.0 mL and a mass of 7.04 g for your first distillate. What is the d...
A: Density - Density is basically the degree of compactnesss of a system.
Q: Predict the formula for the binary compound formed between Cu(II) and P.
A: The elements given are Cu(II) and P.
Q: QUESTION 9 What is the IUPAC name for (CH33CCH2CH,CH3 O 2,2-dimethylpentane O 1-(tert-butyl)-propane...
A: Note: As per our guidelines, we are supposed to answer only one question when multiple questions are...
Q: a) 453 mg to g b) 6.0 x 10-9: m to pm c) 2.5 x 10-2 mm to um d) 3.85 x 10° g to kg e) 0.0065 mm to n...
A:
Q: 223.83 g of mercurous sulfate is dissolved in enough water to form an unknown concentration of solut...
A: Given: Atomic mass of Hg = 200.59 g/mol Atomic mass of S = 32.065 g/mol Atomic mass of O = 15.99 g/m...
Q: A chocolate manufacturing plant produces chocolate coated macadamia nuts. One specific batch of choc...
A: Mass is one of the seven basic physical properties. It measures the amount of substance present. Its...
Q: What results when (Z)-3-methylpent-2-en-1-ol is treated with tBuOOH, Ti(OiPr)4, and (-)-DET? HO an e...
A: Given reaction is known as Sharpless Asymmetric epoxidation reaction. In this reaction , 2,3-epoxyal...
Q: f) 6L of 1mM IPTG. You need to prepare a stock solution of 1M IPTG that will be sufficient to give y...
A: Desired solution is molarity = 1 mM volume = 6 L Stock solution molarity = 1 M molar mass of IPTG ...
Q: Compare the conformational profile of butane (A) with that of 1,2-dibromoethane (B) While the methyl...
A:
Q: 4. Barium sulfate is formed from reacting barium hydroxide and sulfuric acid. Indicate the ion/s tha...
A:
Q: Which of the following statements is FALSE for the chemical equation given below in which nitrogen g...
A: Nitrogen reacts with hydrogen to form ammonia. The equation for the balanced chemical reaction is gi...
Q: Which statement is the best description of the pattern for ionic charge on the periodic table? ...
A: Given that : We have to identify which of the following statements is the best description of the pa...
Q: Name the following molecule by its IUPAC name. Br Cl CI > View Available Hint(s)
A: 1- In the first structure, numbering from Cl carbon side then at C1 and C3 two Cl and at C2 on Br at...
Q: How many C atoms are there in a 41.1 gram sample of C ? atoms
A:
Q: Arrange the following salts in order of increasing lattice energy: CaS, SrI2, MgBr2, NaF, BeO, Fe3N2...
A:
Q: a common preservative used in hematology and blood bank
A: Different types of preservatives are used as per the laboratory requirement. Preservatives used in b...
Q: In order to prepare 100 mL of a 0.50 M sodium phosphate buffer, pH 6.2, how many grams of sodium pho...
A: Given: pH of solution = 6.2 Concentration of buffer = 0.50 M Volume of buffer solution = 100 mL = 0...
Q: Which reagent in cach of the following pairs is more nucleophilic? Explain your answer. NH, or NH, (...
A: As you have asked only B subpart, so, we will solve the same for you. B. Nucleophile is a species th...
Q: Determine whether the mixing of each pair of solutions results ina buffer.a. 75.0 mL of 0.10 M HF; 5...
A: Given options are : a. 75.0 mL of 0.10 M HF; 55.0 mL of 0.15 M NaFb. 150.0 mL of 0.10 M HF; 135.0 mL...
Q: Iron (III) ions and thiocyanate ions react to form a complex ion according to the following equation...
A: The equilibrium reaction taking place is given as, => Fe3+ (aq) + SCN- (aq) -----> FeSCN2+ (aq...
Q: How would you synthesize 3-hexyne using ethane as your only carbon source? You may use any reagents.
A: The question is based on the concept of organic reactions. we have to convert the given reactant i...
How many electrons, protons, and neutrons are there in the ion of the hypothetical element
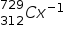
Step by step
Solved in 2 steps with 2 images


