Q: Benzyl bromide (C6H5CH2Br) reacts rapidly with CH3OH to afford benzyl methyl ether (C6H5CH2OCH3).…
A: The first step involves the attack of Br ion via SN1 pathway to form the carbocation. The next step…
Q: Which compound below is most likely to undergo SNAr reaction using NaOCH3 as the nucleophile?
A: In this question, as option c has 2 nitro groups present alongside F. It will be most likely to…
Q: Which alkyl halide in each set is more reactive in an SN2 reaction with hydroxide ion? or Br 오아 or…
A:
Q: Which carbonyl is more suceptible to nucleophilic attack, carbonyl of cyclohexanone or carbonyl of…
A: The two carbonyl compounds given are hexanal and cyclohexanone.
Q: Rank the following in increasing order of reactivity towards nucleophilic acyl substitution a.…
A: The rate of nucleophile attack to the CO group increases, if the C of CO group is electrophilic.
Q: This is Wittig Rxn: Whatever mechanism you choose to draw is fine since you can leave the base as…
A: Wittig reaction is carried out between aldehyde or ketone and ylide to form the alkene .
Q: Arrange the following compounds in the increasing order of their reactivity in nucleophilic attack…
A: More the electrone deficiency on the carbonyl carbon , more easily the attack of the nucleophilie…
Q: Arrange the following reagents in INCREASING order of * nucleophilicity* in protic solvent: NaOCH,…
A:
Q: (a) p-Nitrobenzaldehyde is reactive toward nucleophilic additions than p- more methoxybenzaldehyde.…
A: a)
Q: "Drawing the Product of Inversion in an SN2 Reaction Label the nucleophile and leaving group, and…
A: Nucleophile : It should contained lone pair or negative charges. leaving group : this group will…
Q: Arrange these leaving groups in order of increasing E2 reaction rate.
A: Here among the leaving groups , the weaker the base , more is its ability to leave . They have…
Q: Explain why a methoxy group (CH3O) increases the rate of electrophilic aromatic substitution, but…
A: Methoxy group (CH3O) increases the rate of the rate of electrophilic aromatic substitution reaction,…
Q: What site on the alkyl halide is likely to get attacked by the cyanide (in other words, which atom…
A: Electrophilic sites are those sites which are electron deficient.
Q: Why is the presence of acid necessary in the reaction of carbonyl compounds with certain…
A: Reaction of carbonyl (suppose acetone) with nucleophile like 2,4-dinitrophenyl hydrazine follows…
Q: Choose the most correct set of reagents for the following reaction.
A: Alcohol can be converted into the ether by a nucleophilic substitution reaction mechanism. In the…
Q: Rank the following according to increasing reactivity with 1 as the least reactive, and 4 as the…
A: The reactivity order for nucleophilic acyl substitution is as follows:
Q: What reagents A and B would be most appropriate for the following synthesis? 1. NaOEt, EtOH A CH 2.…
A:
Q: 4. a. Which of the following would be more reactive in an electrophilic aromatic substitution ?…
A: 4 a. Electron donating groups increases the reactivity of benzene ring towards electrophiles.…
Q: Which product (if any) would likely be the major product for the following reaction? a. Neither…
A: Alcohols upon dehydration in acidic conditions to yield an alkene , in this reaction carbocation…
Q: . Like acid chlorides, acid anhydrides react with alcohols in the presence of pyridine to form…
A: We will write the nucleophile, electrophile, base and tetrahedral intermediate. Then we will draw…
Q: a. Increasing acidity: p-Bromobenzoic acid, p-nitrobenzoic acid, 2,4-dinitrobenzoic acid NH2 NH2 ZHN…
A: Answer of this question :-
Q: Consider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and D. (a)…
A: The given tetracyclic aromatic compound is shown below. The presence of electron releasing groups…
Q: Explain why a nucleophilic reagent such as ethoxide adds to an alkyne more easily than it adds to an…
A: The unsaturated hydrocarbons are of two types namely alkenes and alkynes. The alkenes consist of at…
Q: -Cf3 Nucleophilic Attack
A:
Q: What vinylic halide couples with styrene (C6H5-CH=CH2) in order to synthesize each of the following…
A: Heck Reaction: Heck reaction is a coupling reaction in which vinyl halides or aryl halides couple…
Q: Which is the better retrosynthesis for the given target molecule? Explain, and provide a one-step…
A: E option is correct.
Q: Explain Electrostatic potential maps of four halomethanes (CH3X)
A: The given reaction is nucleophilic substituion reaction in which nucleophile substitute the halogen…
Q: Consider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and D. (a)…
A: The compound is
Q: (a) Which of the following phenols is the least acidic? (b) Which of the following phenols is the…
A: In phenols on increasing the electron donating groups like methyl or methoxy at para or ortho…
Q: Which of the following reactant will not favour nucleophilic substitution reaction ? (a) (b) Ph – Br…
A:
Q: Explain the difference in reactivity between CH3OH2 and CH3OH in a nucleophilic substitution…
A: The alcohol contains a two lone pair and therefore, will be reactive for the attack on reagent to…
Q: HBr can be added to an alkene in the presence of peroxides (ROOR). What function does the peroxide…
A:
Q: Consider carbonyl compounds A–E drawn below. (a) Rank A–E in order of increasing stability. (b) Rank…
A: Introduction: The stability of the carbonyl compound can be determined by the electrophilic…
Q: Rank the species below in order of increasing nucleophilicity in protic solvent.
A: Nucleophile are species which have excess of electron density either have filled orbital or lone…
Q: Amines are good nucleophiles, even though they are neutral. How would the rate of an SN2 reaction…
A: The SN2/E2 reaction of alkyl halide is with neutral nucleophile/base, the increasing polarity of the…
Q: Identify the stronger nucleophile in each pair of anions. a.Br− or Cl− in a polar protic solvent…
A: A nucleophile is an electron rich species which consists of lone pair of electron pair or pi bond to…
Q: Which of the following is more nucleophilic and why? CH30 (CH3)3CO 1 2 Compound 2, because its…
A: Strong nucleophile means an atom or a group carry lone pair of electrons. It effectively go to…
Q: Rank the following species from best nucleophile to poorest nucleophile in the given solvent: OH A B…
A: Nucleophilicity is determined by the tendency to donate the electron pair. The species which carry…
Q: (b) Which of the following will react most slowly with cyanide nucleophile (NC-) in an Sn1 reaction?…
A:
Q: Which of the following statements best describes why I- is a better nucleophile than F in solution?…
A: e is correct answer. A and B both offer explanations as to why I- is a better nucleophile than F- in…
Q: d. Hint: the acetylide ion is both a strong base and a strong nucleophile. Br Na+ ECH THF
A: given substrate is a one degree alkyl bromide and it will go elimination and substitution reaction…
Q: Explain the difference in reactivity between CH3OH2 and CH3OH in a nucleophilic substitution…
A: The alcohol contains a two lone pair and therefore, will be reactive for the attack on reagent to…
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. −OH, Br−, F− (polar…
A: The given groups are -OH, Br-, F- To find: The increasing order of nucleophilicity
Q: Arrange in decreasing order according to their reactivity towards an electrophile. H A В
A:
Q: Complete the attached problem
A: A chemical reaction is symbolic representation of the conversion of substances to new substances. In…
Q: Like acid chlorides, acid anhydrides react with alcohols in the presence of pyridine to form esters.…
A: Electrophile is electron deficient species whereas Nucleophile is electron rich species
Q: 1. Rank the following according to increasing reactivity (1 - least reactive, 4 - most reactive in a…
A:
Q: 1. What is the nucleophile in the reaction? 2. The reaction above follows what mechanism? 3. What is…
A: It is the basic part of the reaction mechanism. Here we have to be clear about the nucleophile,…
Q: (c) What is the solvent effect on the SN1 and SN2 reactions?', (d) Which reactant in each of the…
A: In SN1 carbocation intermediate is formed.. And in SN2 transition state is formed..
B only
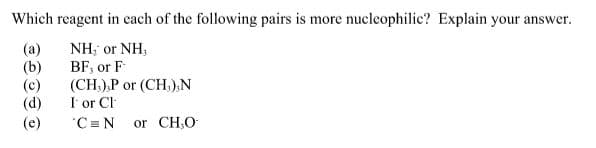
Step by step
Solved in 2 steps

- Arrange the structure on the image with regard to the reactivity towards hydrolysis. 1 being the least and 3 being the mostWhen drawing organic chemistry mechanisms with NaOCH3, do we automatically dissociate it into Na+ and OCH3-, making it easier to go through SN2 and/or E2 reactions? Does NaOCH3 dissociate, and is this why it is a strong nucleophile?A student was given from the list of the compounds below A, B and D blindly and asked to identify them all. He treated each of them with Brady's reagent (2,4-ditrophenylhydrazine) and isolated a bright yellow compound for one of them, but the other two gave false negatives. The student reasoned that the false negatives may be due to sterics and, on further thinking, it dawned on him that he might be able to rule out one of the false negatives with the haloform test. What compound did he find compatible with the haloform test? That compound did indeed give a false negative in the Brady test. Which of the other two was positive in the Brady test? A = haloform B = Brady A = haloform D = Brady B = haloform A = Brady B = haloform D = Brady D = haloform A = Brady D = haloform B = Brady
- Explain the Summary of Alkyl Halides and SN1, SN2, E1, and E2 Mechanisms ?Draw the products formed by the following acid-base reaction without consideration of the equilibrium position: You do not have to consider stereochemistry. You should include all products. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Separate structures with + signs from the drop-down menu.Suggest reactivity of compound A, B and C in increasing order of E2 reaction
- Rank A, B, and C in order of increasing SN1 reactivity.When the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.Draw a reasonable mechanism for the reaction of E-cinnamic Acid with pyridinium tribromide,using wedge and dash bonds. Provide the stereochemistry of the product.
- Compelling evidence for the existence of a tetrahedral intermediate in nucleophilic acyl substitution was obtained in a series of elegant experiments carried out by Myron Bender in 1951. The key experiment was the reaction of aqueous −OH with ethyl benzoate (C6H5COOCH2CH3) labeled at the carbonyl oxygen with 18O. Bender did not allow the hydrolysis to go to completion, and then examined the presence of a label in the recovered starting material. He found that some of the recovered ethyl benzoate no longer contained a label at the carbonyl oxygen. With reference to the accepted mechanism of nucleophilic acyl substitution, explain how this provides evidence for a tetrahedral intermediate.Compelling evidence for the existence of a tetrahedral intermediate in nucleophilic acyl substitution was obtained in a series of elegant experiments carried out by Myron Bender in 1951. The key experiment was the reaction of aqueous−OH with ethyl benzoate (C6H5COOCH2CH3) labeled at the carbonyl oxygen with 18O. Bender did not allow the hydrolysis to go to completion, and then examined the presence of a label in the recovered starting material. He found that some of the recovered ethyl benzoate no longer contained a label at the carbonyl oxygen. With reference to the accepted mechanism of nucleophllic acyl substitution, explain how this provides evidence for a tetrahedral intermediate.what would a detailed step-by-step mechanism be for a reaction between bromine and trans-stilbene if the solvent, ethanol, is a stronger nucleophile than the bromide ions?

