Q: 750 g potassium iodide 237 mL per bottle Potassium 950 g elemental iodine 1.5 kgs potassium…
A: A numerical problem based on stoichiometry, which is to be accomplished.
Q: Active Ingredient Raw Materials Actual Yield Formulation Dosage Form Packagin Precipitated sulfur…
A: A common ingredient found in pesticides and insecticides is calcium polysulphide . A well known…
Q: A Volumetric Analysis A. Standardization of a Sodium Hydroxide Solution Part A.3. Approximate [NaOH]…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 4. Which
A: Steps in the method of analysis are - => Selection of method : First we select the analysis…
Q: A 0.6447 g portion of manganese dioxide was added to an acidic solution in which 1.1402 g of a…
A: Given data, MnO2 taken = 0.6447 g Chloride sample taken = 1.1402g MnO2 recovered = 0.3521g.
Q: The following mixtures are completely mixed to form mixture D: Mixture Mass (kg) % NaCI % KCI % H2O…
A: The solution is given in below image file: From the given information, It is know that mass of each…
Q: [OH-] = 5.0 × 10–2 Express the concentration to two significant figures and include the appropriate…
A: Consider the given information is as follows; OH-=5.0×10-2H3O+ = ?Acidic, basic or neutral = ?…
Q: 2. What is the gravimetric factor (a) For acquiring the weight of Ag:CrO, from the weight of AgCI?…
A: The analytical method that is employed for the quantitative determination of unknown substances in…
Q: Can you please fill the rest of tghe data based on the on inforamtion you have and answer Average…
A: Calculations for trail 1:
Q: To prepare standard KHP ( M.W.= 204.22 ) solution for the KHP lab, you weighed out 0.9539 ( ±…
A: The balanced chemical equation has the number of atoms of the reactants becomes equal to the number…
Q: The toxicity of DDT (C14H9Cl5) led to a ban on its use in the United States in 1972. Determinations…
A: We have to predict the concentration of DDT .
Q: ?Al+?HCl → ? AIC3+?H Which set of coefficients wil balance this equation?
A: Steps involved: Al balancing Cl balancing H balancing
Q: 4. Balance: K + H,O à КОН + H,
A:
Q: Using dimensional analysis, find the theoretical mass of the Zn3(PO4)2-4H20 precipitate that should…
A: Given : Mass of Zn(C2H3O2)2.2H2O reacting = 2.19 g Molar mass of Zn(C2H3O2)2.2H2O = Atomic mass of…
Q: Predict the products and balance: Mg(NO_3)_2 +Na_2CO_3 ->
A:
Q: When the high-temperature superconductor yttrium barium copper oxide is heated under flowing H,, the…
A: A superconductor is a substance that conducts electricity without resistance when it becomes colder…
Q: From 0.8 av lb of 20% w/w KOH solution, O72.6 121 142 86.2 g of 12 percent w/w KOH may be produced.
A: A question based on dimensional analysis that is to be accomplished.
Q: What volume of 1.50 MSrCl2 is needed to prepare 525 mL of 5.00 mMSrCl?
A: 5.00 mM=0.00500M Here we use the formula = M1V1=M2V2
Q: ___ NH3 + ___ O2 → ___ NO + ___ H2O balance the equation
A: Balancing the reaction is to make count of each atom equal on both sides. To balance atoms…
Q: The mercury in a 1.1113-g sample was precipitated with an excess of paraperiodic acid, H5IO6: 5Hg2+…
A: Soln
Q: 5-) 1,200 grams of aluminum of an impure NH4AI(SO4)2 sample is converted into Al203 by precipitation…
A: Given data contains, Mass of NH4Al(SO4)2 is 1,200 grams. Mass of Al2O3 is 0.1798 grams. (a) The mass…
Q: Balance the following equation and charges, Cr2O7^-2 + Fe^+2 + H2O --> Cr^+3 + Fe^+3 + OH^-
A:
Q: 3. Complete the following reactions (indicate product(s) and balance). А. НBr + LIOH В. Н2SO4 + КОН…
A:
Q: n the textile industry, chlorine is used to bleach fabrics. Any of the toxic chlorine hat remains…
A: Given values -> Weight of Na2S2O3 = 146.66kg =146660gram Weight of Cl2 = 42.09 kg = 42090gram
Q: pl•K) AGO
A:
Q: Na oH 4. solid copper(II) carbonate is heated oules tranred durig 0.22 bas (HOh2y0.21 5. solid…
A: Here we have to write the balanced chemical equation and net ionic equation for reactions in…
Q: 2NO2(g) N204(g). Kc = 0.212 the reaction proceeds more to the right True False
A:
Q: Q14. For the emerging sodium-ion batteries, there are a number of cathode materials under…
A: Theoretical capacity: Qtheoretical = (nF) / (3.6*Mw) mAh g-1..........(1) Molar mass FeOF = 90.8…
Q: -2 Molarity of Standard Ag(NO), 5.2167/10 M Trial I Trial II Trial III Mass of Unknown 0.237 o. 232…
A: The data given is,
Q: d) 250 mLof o-0750 M Ag NO3 from the solid reagert -
A: In multiple questions we solve only first question according to the Bartleby guidelines. Calculate…
Q: Balance the eauations below: 11) P + _O2→ P205 12) Na + H20 > NaOH + _H2 13) Ag20 → Ag + O2
A:
Q: %purity of KHP: 99.80% FORMULA WEIGHT of KHP: 204.22 g/mol STANDARDIZATION OF NAOH SOLUTION TRIAL 1…
A: Reaction between acid and base is termed as neutralization reaction, It leads to formation of salt…
Q: Balance CH2O + KMnO4 = _______________________ under neutral conditions
A: The balanced chemical reaction is
Q: Is this equation balanced! AgNO3 + 2 KCI -->ABCI + KNO3 O Yes O No
A:
Q: How many mililiters of 0.1126 M Ba(NO3)2 contain 11.45 g of Ba(NO3)2
A: Given : concentration of Ba(NO3)2 = 0.1126 M And mass of Ba(NO3)2 = 11.45 g Molar mass of Ba(NO3)2 =…
Q: Considere la siguiente reacción de neutralización entre un ácido fuerte y una base fuerte (ecuación…
A: First Concentration of acid can be calculated using mass and volume . Then using balanced reaction…
Q: . What is the mass in grams of potassium dichromate(K2Cr2O7) are required to prepare a 350 cm3…
A: We need to find mass in gram of potassium dichromate. Volume of solution = 350cm3…
Q: Calawate the number of grams Mgl6H)2 necded To mace 550 ml or 0,420 M COH)2 SOlunon? im olu
A:
Q: Calculate the mass of Na2S203.5H20 needed to prepare 250 cm3 of a 0.011 M solution in a volumetric…
A:
Q: Mass of Na2CO3.H2O (g) = 2.12g (g) Mass of the CaCl2.2H2O (g) = 1.98g Mass of the top funnel +…
A: Given data, Mass of Na2CO3.H2O=2.12gMass of CaCl2.2H2O=1.98gMass of CaCO3…
Q: The indicator of Moher regent is. О Кcro4. KCrO4. O KMN04.
A: What indicator used in Moher reagent ?
Q: 1. A 0.1430 g sample of copper was dissolved in 20 mL of 6 mol/L HNO3 and diluted to 100.0 mL in a…
A: The concentration of the standard solution in terms of g/L is 0.0143 g/L. Since the intercept on the…
Q: 1. H DCC 2. LIAIH4 then H30* HO,
A: In the above reaction, Carboxylic acid with DCC (dicycohexydicarodimide) and amine to give amide and…
Q: State whether the following analysis is quantitative or qualitative analysis or both. i)…
A: In qualitative chemical analysis, we identify whether a chemical substance of interest is present in…
Q: Calcium stearate (Ca(OOC(CH2)16CH3)2) is sometimes used as a lubricant in the processing of…
A: Given: Sample of PVC contains = 2 wt% calcium stearate Mn = 25000 To calculate: Mn of balance of the…
Q: 3. In a given dilution, the volume of the solution increases from 10.00 mL to 50.00 mL. We can…
A: The number of moles remains constant. Let the molarity of original solution be M1 and the molarity…
Q: 0.500 cm3 of concentrated H2O2 solution diluted exactly to 50.00 cm3 by water. 10.00 cm3 samples…
A: Answer :- Molarity (mol/dm3) of original concentrated H2O2 sample = 9.09 mol/dm3 Mass…
Q: Is this equation balanced: AGNO3 + 2 KCI -->ABCI + KNO3 O Yes O No
A:
Q: What are the coefficients necessary to balance nahco3
A:
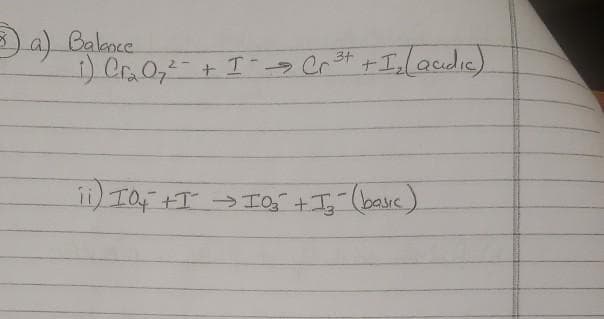
Step by step
Solved in 2 steps with 1 images

- If an Ecell of 0.495V was noted when your cell was set-up. What is thesolution pH?A pH probe/meter uses the following equations: Ecell = L + 0.0592 log a1 = L - 0.0592 pH Where L = L1 + EAg/AgCI + Easy= constants L1 = - 0.0592 log a2 a1 = activity of analyte solution a2 = activity of internal solution Questions: How will measured pH value be affected vs “real” pH if the temperature of the sample is 30C when pH was measured? How will measured pH value be affected vs “real” pH if HCl in pH electrode, became 0.15M instead of 0.1M? How pH value will be affected vs “real” pH if the glass of the pH electrode is not fully hydrated? Please answer all questions and provide a brief explanationA 0.53 g sample containing KBr (MW=119) is dissolved in 50 mL of distilled water. Titrating with 0.04614 N AgNO3 requires 28.5 mL to reach the endpoint. Calculate the %w/w KBr in the sample. (Round-off your answer to 2-decimal places) Answer = ____%
- 3I- + OCl- + 2H+ ----> I3- + Cl- + H2: A 25.00 ml sample of liquid bleach was diluted to 1000 ml in a graduated flask. A 25 ml portion of the diluted sample was pipetted into an Erlenmeyer flask and treated with excess KI to oxidize OCl- to Cl- and I3- was produced at the end of the reaction. The released I3 was titrated with 0.09892 M Na2S2O3 and 8.96 ml was spent to reach the turning point besides the starch indicator. What is the percent by weight/volume of NaOCl in the bleach sample? (NaOCl:74.44 g/mol)1. 1093-g sample of impure Na2CO3 was analyzed by residual precipitimetry. After adding 50.00 mL of 0.06911 M AgNO3, the sample was back-titrated with 0.05781 M KSCN, requiring 27.36 mL to reach the endpoint. The percentage Na2CO3 (MW = 106.0 g/mole) in the tested sample is ________ % ? Note: Express final answer using least number of significant figures. 2. The alkalinity of natural waters is usually controlled by OH- (MW = 17.01 g/mole), CO3-2 (MW = 60.01 g/mole), and HCO3- (MW = 61.01 g/mole), which may be present singularly or in combination. Titrating a 10.0-mL sample to a phenolphthalein endpoint requires 38.12 mL of a 0.5812 M solution of HCl, and an additional 18.67 mL of the same titrant to reach the methyl orange endpoint. The composition of the sample is _________% CO3-2 and ___________ % OH- Note: Express final answers using least number of significant figures.Answer w full solution
- You have Falcon tubes containing 10 ml and you should add ,that you have in a voltage series, from which concentration in the dilution series you should pipette 0.1 ml to reach the final concentration of 10-5M in the organ bath Group of response options 10-5M 10-4M 10-3M 10-2M2.1 Calculate the pH of 1.0 M HC2H3O2 solution, using Ka = 1.7 × 10-5 . Begin this calculation by defining the variable x. List the pH answer as “theoretical pH” on the report sheet. Have the instructor check this answer and all subsequent calculations. To test your prediction, obtain about 35 mL 1.0 M HC2H3O2 and add to a clean, dry 50 mL beaker. Measure the pH of this solution. Be sure the electrode is properly submerged and gives a stable reading. Record the pH reading in the “Observed pH” column on the report sheet. The value should be within ± 0.2 pH units of the calculated value. From the experimental pH and beginning concentrations of the solution components, calculate the remainder of the entries across the table to pKa . Save this solution for Sections 2.3 and 2.6. 2.2 Calculate the pH of 0.10 M HC2H3O2 solution. List this answer as “theoretical pH” on the report sheet. Test your prediction by placing 5 mL of 0.10 M HC2H3O2 in a test tube and testing its…Question 2 (b) Name and discuss five stages in which gravimetric analysis procedure may be divided
- Following the monograph procedure, a 724-mg of aspirin (MW=180 g/mol) dissolved in 18.5 ml of cold neutralized alcohol. This solution was then initially titrated with 0.101 N sodium hydroxide solution, then later neutralized with 0.104 sulfuric acid. What is the percentage purity of the sample? 1. What is the milliequivalent weight consumed by the acidic titrant? a. 1.5392 g-meq b. 2.0907 g-meq c. 1.4948 g-meq d. 2.1528 g-meq 2. What is the milliequivalent weight consumed by the basic titrant? a. 5.8656 g-meq b. 1.5392 g-meq c. 5.6964 g-meq d. 1.4948 g-meq 3. What is the difference of milliequivalent weight consumed in the reaction? a. -4.1572 g-meq b. 4.3708 g-meq c. 0.5515 g-meq d. 4.1572 g-meqPlease Answer my question from the picture im send... And note With same dengan.. God bless you .. Answer question from CO(SCN) until AgNO3... Thanks from fe2+ until Zn(NH3) me have solutionC8This is for my reviewer please help me with the step by step solution and answer, thank you





