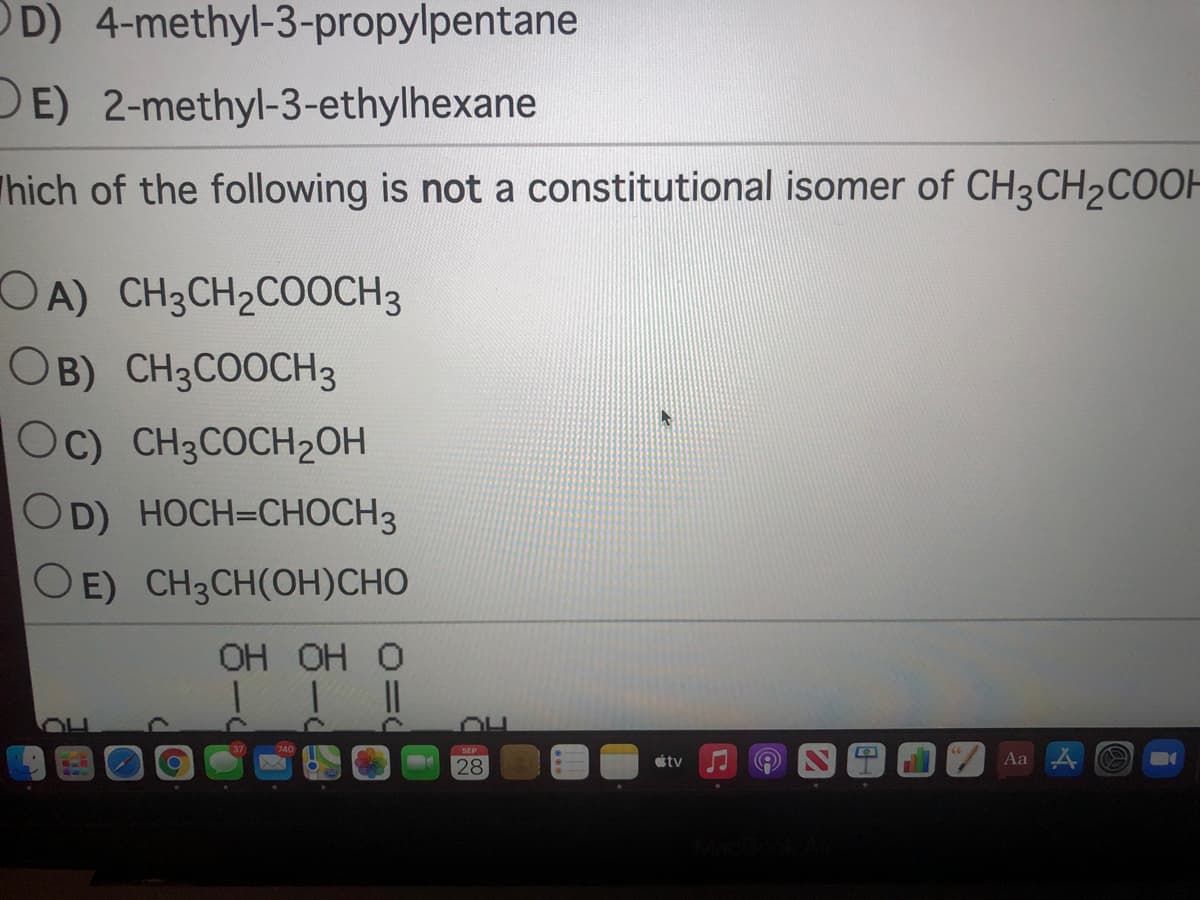
DE) 2-methyl-3-ethylhexane hich of the following is not a constitutional isomer of CH3CH2COOH OA) CH3CH2COOCH3 OB) CH3COOCH3 Oc) CH3COCH2OH OD) HOCH=CHOCH3 O E) CH3CH(OH)CHO OH OH O
Q: 2 Cl2 → A HOʻ C=C-H 2 HBr d) H,SO4 A + B 1) NaNH, (both are alkenes) 2) `Br Na ÔCH3 ..C/ A…
A: Organic reaction mechanisms
Q: Q:- write down the following vehicle designations a) cH3- CH-CH-CH =CH==cH CH3 CH3-CH-CH2-cHE CHECH3…
A: Given,
Q: Directions: Name the following hydrocarbo 1. CH3 CH3 H3C-CH-CH-CH2-CH3 CH3 CH3 2. CHз — с- CH2 CH…
A: Rules for IUPAC naming :- Select the longest chain which contains carbon-carbon single bonds.…
Q: Place the methyl group and hydrogen atom on the structures below to depict the conformation of cis-…
A: Here, we have to place the methyl group and hydrogen atom on the given structures to depict the…
Q: Please do the following . 2-butyne + HgSO4, H2SO4 and H2O 2-methyl-1,3-butadiene + HC ≡ C –…
A: Note: Since you have posted multiple independent questions in the same request ,we will solve the…
Q: Which of the following structures is correct for 3-ethylheptane? CH3 H3C H3C CH3 CH3 CH3 H3C H3C CH3…
A: Here we are asked to write the correct structure of 3-ethylheptane.
Q: Which of the following structures represents trans-1,4-difluorocyclohexane? F F I. II. III. IV. V.…
A: Chair conformation of 1,4- difluorocyclohexane
Q: owing molecules: a- Choloropropane b- Butane
A: Eclipsed form is if dihedral angle is 0° between group If dieheadral angle is 60° then it is…
Q: H₂C-i-OH он H3C-C-CH3 CH3 HC-CH-C=c-CH-CH2-CH CH₂ CH₂ CH CH CH₂-CH₂-CH₂-CH₂-CH₂…
A:
Q: When bromo cyclo-butane interacts with Mg then H20 , it gives cyclo-butane IO butene .II O…
A: The question is based on the concept of organic reactions. We have to identify the product formed.…
Q: Enter the IUPAC name of the following compounds: CH3 CH,CH3 d) CH3 e) CH3-CH(CH3) - CH2-CH, -CH C =…
A:
Q: HaC OH The alcohol above can be classified as and is named secondary, cyclopentanol O tertiary,…
A:
Q: Give the IUPAC name of the following: la) CH3 CH3 (b) CO2H (c) Br CHCH2CH2CHCH3 Br H3C CH3 (d) Br…
A: The IUPAC name of the following compounds has to be given below. (a) (b) (c) (d) (e) (f)
Q: Which of the following has the greatest solubility in CH3CH2CH,CH2CH2CH3? Select one: O a. CH3OH O…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: By considering viewed through the C-2-C-3 bond, which below conformations of 2.3- dibromabutane show…
A: Conformation- III Most stable as it is a Staggered Conformation and All groups are anti to each…
Q: Classify the following compounds as E/Z- structures CH3 HO-CH2-CH2 CH2CHCH2OH H2N CH2-CH3 H2N CH=CH2…
A: For abC=Ca'b' system E/Z nomenclature occurs and E/Z nomenclature is based on CIP rule which states…
Q: All but one of the following conformational models are representative of 2,3-dimethylpentane. Which…
A: Given molecule is, 2,3-dimethylpentane.
Q: 2. Decide whether to use E/Z nomenclature or cis or trans (or if neither are applicable). Then, draw…
A:
Q: Name the following, including cis or trans: a CH;-CH2 H. H CH2-CH2-CH; b Br d Name the following,…
A: Name of the structure with their trans or cis configuration / E or Z configuration is given below.
Q: HOOH HC NBS Light, CC BH3 3-Ethyl-1-isopropyl-cyclahexene IV H202 OH Ag(OAc NaBH4 МСРВА -VI CH,C2…
A: This Reactions are based on the Alkenes which show normally addition Reactions.
Q: CI Ме H. Me CI and H- -H H3C CH3 ČH3 Ме and CH3 HFOH CH3 HỌ -H -OH H- -ОН HO-H ČH,CH3 ČH,CH3 G. F F…
A: The gives molecules are such that one is in Newman projection and the other is in Fischer…
Q: H3C CH2CH3 H. CH,CH3 Но Br H3C HO. Br CO,H -H- CH2CH3 H- OH Но CO,H CH,CH3
A:
Q: 1) Write Ahe condensed structuralformula for the follo wing organie compound. b)', 3 ,5- richoloro…
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost…
Q: Which of the following does not represent 2-methylbutane? H₂C-CH₂ CH3 CH₂ CH₂ CH₂ CH (CH₂)₂ 2 C B D
A: Given,
Q: CH3 CH3 `CH CH, CH, H3C-CH2 -ĊH-C-CH2-CH-CH-CH2-CH2-CHg CH3 CH3 CH2 CH3
A: Given, Hydrcarbon with chain of ten Carbon atoms. Numbering should be done following the IUPAC…
Q: I Enter the IUPAC name for the following alkynes CH3 CH3CHC CCHCH3 (a) CH3 (c) CH3 CH3CH2CC…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Draw curved arrows to show the movement of the electrons that result in formation of the given…
A: a.-OH acts as a base and it has tendency to abstract proton. The curved arrows that show the…
Q: (Specify (E)/(Z) stereochemistry, if relevant, for straight chain alkenes only. Pay attention to…
A:
Q: By considering viewed through the C-2–C-3 bond, which below conformations of 2,3- dibromobutane show…
A: Most Stable conformation is - III As all the Respective groups are anti to each other (Staggered…
Q: Vhich of the following is not a constitutional isomer of CH3CH2COOH? OA) CH3CH2COOCH3 OB) CH3COOCH3…
A: The compound given is CH3CH2COOH.
Q: (a) OH OH (b) OH (c) Но CH3CHCH,ĊHCHCH3 CH2CH2CCH3 -CH3 ČH3 ČH3 CH3
A: In organic chemistry, chemical compounds are mainly formed using carbon, oxygen, hydrogen and…
Q: What is the IUPAC name for the following molecule? O (2E,5Z)-2-ethyl-5-methylhepta-2,5-diene O…
A: The first and the foremost step while naming the IUPAC name of an organic compound is the…
Q: ? Benzene D-N(CH3)2 D-OCH2CH3 O-CO,H O-CH2CH3
A:
Q: A 1,2-cis disubstituted cyclohexane, such as cis-1,2-dichlorocyclo- hexane, must have one group…
A: Stereoisomers are different from constitutional isomers. They not only have the same molecular…
Q: 2)- Determine the total number of conformers of the tetra-substituted cyclohexane structure given…
A: Hello. Since your question has multiple sub-parts, we will solve only the first three sub-parts for…
Q: Classify the following reactions as addition, elimination, substitution or rearrangement: CH3CH3 +…
A: Given reactions are : Whether these reactions are : Addition, Elimination, Substitution or…
Q: Classify the following organic reactions
A: Given reactions,
Q: CH, CH3 CH2-CH3 CH,-C-CH-CH-CH2 CH3 CH2-CH-CH, CH3 d) CH2-CH3 CH;-CEC-CEc-CH-CH-CEc-CH3 CI e) CH3…
A: Since you have posted multiple subpart question, we will answer the 3 subparts (d,e,f) for you.…
Q: (a) Name the compounds whose structures are shown below: CH-CH-CH3 či CH (ї) Н (ii) CH,CH H2C=C-CH…
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: choose the correct name for the attached diagram (2E, 6E)-3-ethyl-7-methyl-2,6-dien-1-ol C. (2Z,…
A: IUPAC name of the organic compound is composed of the following three components:Root name = depends…
Q: hsidering structures below, which of the following statements is true? 유 1 H-C-O-CH₂2 CH₂OH ||…
A: we have to identify the correct statement with respect to the given structures
Q: 36. Which of these formulas is(are) not valid for hexane? A. CH14 B. C6H6 C. C6H16 D.…
A: Hexane is a saturated open chain hydrocarbon The general formula for those is CnH2n+2 hexane number…
Q: CH,Br CH Br 十 Br Br H- H- -Br Br Br Br ČH3 CH3 YZA BCD EFG VWX
A: Assign the priorities to the different groups attached to the chiral carbon as per…
Q: 7) Fill in the Newman Projections with the carbon chain from the molecule below in the Anti-, Gauche…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Give the complete IUPAC name (with E/Z designation) of the following alkynes and aromatic compounds.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: By considering viewed through the C-2-C-3 bond, which below conformations of 2,3- dibromobutane show…
A: In alkanes, due to free rotation around carbon carbon single bond infinite number of structure are…
Q: ider three molecCules in the image below. Select t wo statements that are correct. H CH3 :0: H H3C…
A: Forces that act between the molecules are known as molecular forces. like dipole-dipole, induced…
Q: Conversion of Compound into complete structure. A.CH3(CH2)8CH3 B. CH3(CH2)4OH C. CH3CCl3 D.…
A: The given compounds has to be converted into its complete structure.
Q: When bromo cyclo-butane interacts with Mg then H20 , it gives .I суclo- butane .II сyclo- butene…
A:
Step by step
Solved in 2 steps with 2 images

- 1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the aboveA chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactionsArrange the following groups in order of increasing priority. Q.) -CH3 -CH2OH -CH2NH2 -CH2Br
- Alcohols are important for organic synthesis, especially in situations involving alkenes. The alcohol might be the desired product, or the OH group might be transformed into another functional group via halogenation, oxidation, or perhaps conversion to a sulfonic ester derivative. Formation of an alcohol from an alkene is particularly powerful because conditions can be chosen to produce either the Markovnikov or non-Markovnikov product from an unsymmetrical alkene. Using your reaction roadmap as a guide, show how to convert 4-methyl-1-pentene into 5-methylhexanenitrile. You must use 4-methyl-1-pentene and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.Consider the molecule: rate the priority functional groups from highest to lowest A. Alkyl chain B. Ketone C. Carbonyl D. AnhydrideDoes the data provide evidence for the claim that the reaction of Br2 with alkenes proceeds via anti‐addition? The mleting point of the product was found to be 234.5-235.5C chemical reaction: trans-stilbene+pyrimindine tribromide --->acetic acid 1,2-di bromo-1,2-diphenylethane product was trans-1,2-dibromo-1,2-diphenylethane
- A hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-yne
- KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanoneName the major product of the reaction between (S)-2-bromohexane and KCH3COO. Be specific with the configuration.I have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:



