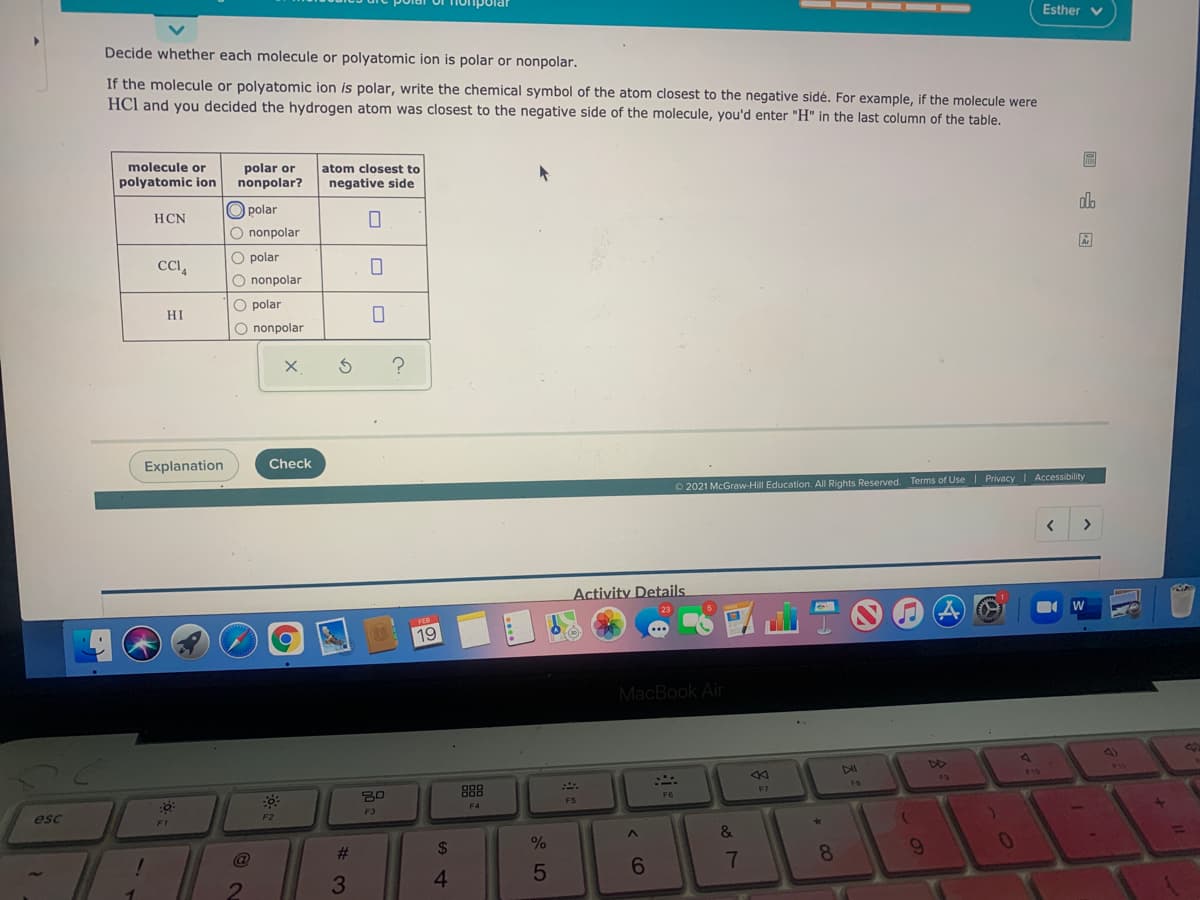
Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative sidé. For example, if the molecule were HCl and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter "H" in the last column of the table. molecule or polar or nonpolar? atom closest to polyatomic ion negative side O polar O nonpolar HCN O polar CCA O nonpolar O polar HI O nonpolar
Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative sidé. For example, if the molecule were HCl and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter "H" in the last column of the table. molecule or polar or nonpolar? atom closest to polyatomic ion negative side O polar O nonpolar HCN O polar CCA O nonpolar O polar HI O nonpolar
Chapter5: Chemical Bonding
Section: Chapter Questions
Problem 46E: CH3COCH3 (acetone) is a common laboratory solvent that is often used in nail polish remover. Its...
Related questions
Question
100%
Can y’all please stop give me wrong answers
Transcribed Image Text:nonpolar
Esther v
Decide whether each molecule or polyatomic ion is polar or nonpolar.
If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative sidé. For example, if the molecule were
HCl and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter "H" in the last column of the table.
molecule or
polyatomic on
polar or
nonpolar?
atom closest to
negative side
O polar
HCN
do
O nonpolar
O polar
CI,
O nonpolar
O polar
HI
O nonpolar
Explanation
Check
O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use | Privacy Accessibility
Activity Details
19
MacBook Air
2)
10
888
F6
F7
F5
F4
esc
F2
F1
&
@
%23
24
8
6.
4.
6
Expert Solution
Step 1

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning



General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning