Q: Draw the products formed when CH3COCH2CH2CH = CH2 is treated with each reagent:(a) LiAlH4, then…
A: a.
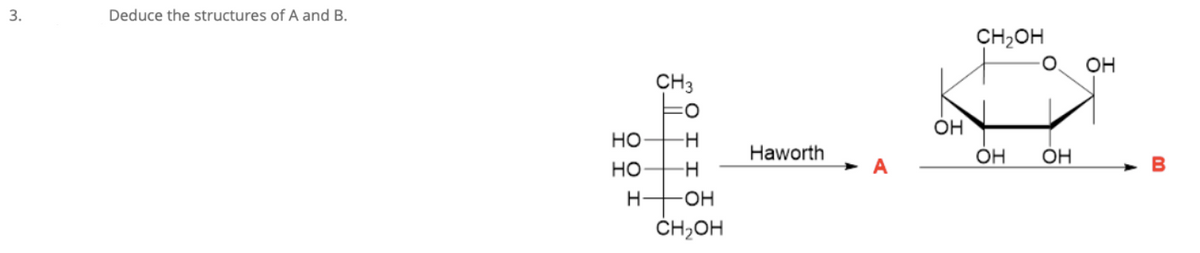
Q: Deduce the structures of A and B. CH2OH OH CH3 ÓH но -H- Haworth ÓH но A B H он ČH;OH
A: In this question, we want to identify the structure of A and B How we can identify the structure,…
Q: 6. CH3 HNO3. H₂SO4 ?
A: In this question we have to tell the product of the reaction
Q: What reagents are needed to convert (CH3CH2)3CCH=CH2 to each of the following compound?
A:
Q: 6. Explain the following acid trends. a. CH3CH2CBr2COOH is a stronger acid than CH3CH2CH2COOH b. H20…
A: ->The species which form stable conjugate base that is most acidic. ->The species which can…
Q: e structure of D-arabinose is shown. CHO НО- -H H-OH |- -OH CH₂OH Which structure is the enantiomer…
A: ->Enantiomer are the isomers which has just reverse configuration at every carbon center.…
Q: Treatment of cholesterol with MCPBA results in formation of a single epoxide A, with the…
A: Stereoselectivity is the property of a Compound or reaction in which a single reactant forms an…
Q: Which of the following is a hydrogen bond donor? A. CH3CH2CH3 B. H2O2 C. H2S D. HBr
A: For any molecule to act as a hydrogen bond donor, it should be having a H atom attached directly to…
Q: 2) Classify the following tannins based on structural properties and circle the components of cach…
A: The aromatic biomolecules that have many phenolic molecules in a systematic way are known as tannin.…
Q: 3. Deduce the structures of A and B. CH2OH OH CH3 Fo Но Haworth OH OH но B -ОН ČH2OH
A: In this question, we want to draw the structure of A and B You can see the structure of A and B…
Q: Classify each substituent as electron donating or electron withdrawing. a. -OCOCH3 b. -CN
A: Electron withdrawing groups are those groups which decreases the electron density on the benzene…
Q: What is the stereochemistry relationship between (2S,3R)-2,3-dibromoheptanoic acid and (2R,3s )-2,3-…
A: Third option. They are enantiomers
Q: O 3. When HOCH2CH2CH2CH2COCH2CH2CH2CH20H is heated in the presence of acid catalyst, a reaction…
A: HOCH2CH2CH2CH2COCH2CH2CH2CH2OH on heating in presence of acid catalyst will undergo cyclization by…
Q: Determine the major product of the rxn CH3CH2OH C6H5CH=CHCCH3 + CN- CH3CO2H CN O Он CN OH А. Ph C.…
A:
Q: When 2-heptine was treated with aqueous sulfuric acid containing mercury(II) sulfate, two products…
A: When 2-heptyne was treated with aq. H2SO4 containing mercury (II) sulphate There are two products…
Q: 1. The label has fallen off a bottle containing a sugar with the formula C6H the unknown sugar at…
A: Glucose is a simple carbohydrate, or sugar. It is one of several kinds of sugars. It is important…
Q: Which of these reagent(s) will not react with HOCH2CH2CH2COOH? A) NaCN in ethanol B) C2H5OH in…
A: NaCN in ethanol reacts with aldehyde and ketone not with an acid or an alcohol.
Q: Draw the products formed (including stereoisomers) when attached compound is reduced with NaBH4 in…
A: Reduction of aldehyde: NaBH4 reduces the aldehyde into a primary alcohol. It acts as a source of…
Q: Identify the structures of C and D in the following reaction sequence.
A: Figure 2
Q: What reagents are needed to convert (CH3CH2)3CCH=CH2 to each of the following compound? a.
A: Oxidation of alkene to ketone means addition of oxygen to alkene and lead to formation of ketone.
Q: Draw and label the chemical structure of the following compounds and label its replacable Hydrogen…
A: KHP is Potassium hydrogen pthalate . We would draw structures and label replaceable hydrogen…
Q: 1. Classify each substituent as electron donating or electron withdrawing LOCH .C(CH3)3 a. OCOCH3 b.…
A: Electron donating group Electron withdrawing group
Q: REFER TO THE SCHEME FOR THE SYNTHESIS OF LIDOCAINE SHOWN BELOW NO2 SnCl,/ HCI LNH3 CI NH2 Кон…
A: The explanation of the organic reaction is given below
Q: OMe OMe H2SO4 2 equiv. OMe OMe
A: Given reaction is : Draw an arrow pushing mechanism and explain why less than two substitutions are…
Q: Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent.a. HClb.…
A: a.
Q: Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH…
A:
Q: 8. Which one of the following compounds is optically inactive (achiral)? Ph.. A В E
A:
Q: 17. Provide the major organic product of t 10 NaOCH3 CH3OH
A: We have given the organic reaction and we have to find the major product of the reaction.
Q: Part I. Formation of the bromohydrin from a-methylstyrene ОН C-CH,-Br A Br CH3 Br2 C=CH2 -c-CH2 CH3…
A: When the hydroxy (-OH) group is added to the carbon that is attached to three other carbons then the…
Q: What product is formed when attached compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4,…
A:
Q: What product is formed when each compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4,…
A: Since among the above reagents , Na2Cr2O7, H2SO4, H2O is a strong oxidising agent Hence they will…
Q: Give the organic product: :Ö-CH;CH; + но-сн-сн; ? CH;CH3 OH II OCH2CH3 H OCH2CH3 "OH ČH2CH3 CH2CH3…
A:
Q: 6. What is the relationship between A and B? HO OH II A) They are constitutional isomers They are…
A:
Q: What product is formed when attached compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4,…
A: Introduction: Tollens reagent is Ag2O, NH4OH. It oxidizes aldehyde to carboxylic acid only. Jones…
Q: indicate the stereochemistry of any chirality centers formed in the reaction. CH;OH H Br NaOH NaH Br
A: Since you have posted a question with multiple sub- parts, we will solve first three sub- parts for…
Q: 1. Starting with diethyl malonate, prepare the compound shown. All H₂C CH₂ H₂C -CH₂
A:
Q: Describe a synthesis for each CH3 CH2CHz NHa the following compound of CH3 CH = CH ₂
A:
Q: C12H1602 OH O H. 1) NaOEt, ELOH Br 4 possible products with unique carbon skeletons (not counting…
A: Detail mechanistic pathway is given below to find out the other products
Q: Draw the indicated starting material and fill in the missing products or reagents (A – D) to…
A: Number of functional group is associated with organic compounds which impart specific chemical and…
Q: What product is formed when D-Gulose is treated with a. CH3I, Ag2O b. The product in (a), then…
A: D-gulose is a aldohexose sugar . it is a monosaccharide which is found very rarely . reactions of D-…
Q: What product is formed when each compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4,…
A: Since among the above reagents , Na2Cr2O7, H2SO4, H2O is a strong oxidising agent Hence they will…
Q: Draw the organic product of the following reaction. CH=CH2 Cl2, H20
A: since you have posted multiple questions in a single session, we are entitiled to answer only first…
Q: V. Give the product of the following SN1 reaction: CH3 H,O H3CH2C+E ČH,CH2CH3 to -Br a. Assign the…
A: V. Product of the given reaction,
Q: What reagents are needed to convert (CH3CH2)3CCH=CH2 to each of the following compound? а. b. CI CI…
A:
Q: What sugar units make up the structure shown? CH„OSO;- НО H H ÇOO- H NHCCH3 H H. K ÖH H он O…
A:
Q: Which of the following is the best representation of an aqueous lysine ? CH2 CH2 CH2 CH2 H,N-Ç-COOH…
A:
Q: Threonine is a naturally occurring amino acid that has two stereogenic centers. a. Draw the four…
A: a) The four stereoisomers of threonine are -
Q: Propicnic acic Is treated first with Br2 and one equivalent of PB 3, then with a large excess of…
A: Given
Q: What reagents are needed to convert (CH3CH2)3CCH=CH2 to each of the following compound? а. b. CI CI…
A: The solution is as follows:
help me
Step by step
Solved in 3 steps with 2 images

- The following compound readily eliminates CO; to form a conjugated six membered ring. (a) Complete the reaction with drawing the possible structure and (b) explain why this reaction goes steadily.(A) Provide the major organic product for the reaction below (B) Would the product be optically active of optically in active?deduce the type of each reaction below:
- 1. The of alka zeltzer in iced water will give a result and observation of _________. alka zetltzer dissolved readily alka zeltzer partially dissolved No reaction 2. In the Le Chatelier's Principle, Co(H2O)6 with oxidation of +2 is _________ in color. blue violet pinkSuppose you have compounds A–D at your disposal. Using thesecompounds, devise two different ways to make E. Which one of thesemethods is preferred, and why?Rank the attached compounds in order of increasing stability.
- 1. Explain how you could synthesize butane. Be sure to list all reactants and reagents required. 2. Explain how Hammond's postulate accounts for the higher selectivity of bromination reactions as compared to chlorination reactions.What is the major organic product obtained from the following reaction? A, B, C, or D?Provide the neccessary reagents next to the arrows



