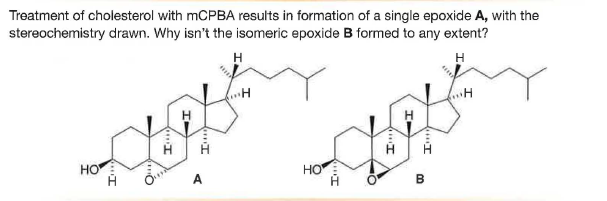
Treatment of cholesterol with MCPBA results in formation of a single epoxide A, with the stereochemistry drawn. Why isn't the isomeric epoxide B formed to any extent? H HO Но A HI
Q: Triacylglycerol L yields compound M when treated with excess H2, Pd-C. Ozonolysis of L([1) O3; [2]…
A:
Q: Draw the products of attached reaction and indicate stereochemistry aroundstereogenic centers.
A: Given: To draw the product and indicate the stereochemistry of the reaction,
Q: 6. Explain the following acid trends. a. CH3CH2CBr2COOH is a stronger acid than CH3CH2CH2COOH b. H20…
A: ->The species which form stable conjugate base that is most acidic. ->The species which can…
Q: The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form…
A:
Q: Treatment of cholesterol with mCPBA results in formation of a single epoxide A, with the…
A:
Q: 18) Predict the product. ů CH₂CH3 Brz CH,COzH
A: Halogenation of the ketone is the type of the organic reaction in which ketone react with halogen in…
Q: 3. Deduce the structures of A and B. CH2OH OH CH3 Fo Но Haworth OH OH но B -ОН ČH2OH
A: In this question, we want to draw the structure of A and B You can see the structure of A and B…
Q: 3. Propose a mechanism for the reaction on the following page: -CH3 CH3 CI+ in presense of…
A: The mechanism of the reaction is shown as:
Q: All of the OH groups of the β-pyranose form of d-xylose are equatorial. Which of the following is…
A: To find: Beta-furanose form of d-xylose
Q: Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure.…
A:
Q: Ammonia and amines react with epoxides with the same stereospecificity as anionic nucleophiles. Draw…
A:
Q: What is the E2 major product of the structure?
A: The E2 reaction is a bimolecular elimination reaction that accompanies a one-step mechanism.…
Q: The attached isomerization reaction, drawn using D-glucose as startingmaterial, occurs with all…
A: The first step is the abstraction of proton from the carbon atom of aldehyde group to form enolate.…
Q: rehalose is a disaccharide that can be obtained from fungi, sea urchins and insects. Acid hydrolysis…
A: The compound trehalose is types of the disaccharide that is not the reducing sugar. They are…
Q: Draw the structure of the products formed when the sugar below is reacted with sodium borohydride…
A:
Q: H CH₂ H CH3 E -lect one: The ring closure is disrotatory and the two methyl groups are cis in the…
A:
Q: 10. Which of the aldopentoses below will give an optically active aldaric ecid (di-ncid) on…
A:
Q: Identify the organic functional group(s) of the reactant, the reaction type, and predict the…
A: The carbohydrate given contains five carbon atoms and aldehyde group. This carbohydrate is…
Q: 17) Complete the following reaction and name each product with the R and S configuration + CH₂CH3…
A: Here we have to determine the R and S stereoisomers of the product formed in the following given…
Q: 5. Structures A and B are best described as (pte) a) C3 epimers. b) Enantiomers. c) C4 epimers. d)…
A: I solved 5 6 7 questions...if you want more please repost the question with numbers... Note:…
Q: Draw the organic products formed in attached reaction, and indicate the stereochemistry of products…
A: Organic reactions are reactions involving organic compounds. The types of reactions seen in organic…
Q: 5) putting the least acidic first. Rank the following compounds in order of increasing acidity,…
A: Inductive effect: Partial displacement of sigma electrons towords more electronegative atoms side…
Q: Identify the lettered compounds in each reaction sequence.Draw the product formed when…
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Triacylglycerol L yields compound M when treated with excess H2, Pd-C. Ozonolysis of L ([1] O3; [2]…
A: The structure of compound M is,
Q: 7. Draw the structure of the product formed when the sugar below is oxidized using nitric acid to…
A:
Q: 1.) Propose the biosynthesis of 5-methylorsellinic acid: CH3 но. CH3 CO,H
A: Orsellinic acid is o-orsellinic acid which is a phenolic acid. It is of importance in the…
Q: trehalose is a disacharide that can be obtained from fungi sea uchins and insects. acid hydrolysis…
A: From the given information, The hydrolysis of trehalose results the 2 D-glucose molecules, the…
Q: Identify the lettered compounds in each reaction sequence.Draw the product formed when…
A: Intermediate that formed when C6H5CH2CN is treated with the DIBAL-H is;
Q: 3 Trehalose is a disaccharide that can be obtained from fungi, sea urchins and insects. Acid…
A:
Q: OMEM OMEM но.. но. ÓBn ÓBn A 1. I2. NaH CO3 2. B2CI, DMAP OMEM BzO, Which diastereomer A or B gives…
A:
Q: What are the R/S configurations of the sterogenic centers in this molecule?
A: In any molecule, a stereogenic center is a point of location at which interchange of any two groups…
Q: 48. What product is obtained from hydroboration-oxidation of 1-heptyne? OH (A) (B) 49. What is the…
A: The first reaction is the hydroboration oxidation reaction of the alkyne . In this hydroboration…
Q: CH2OH OCH3 OH Но 2. OH a. Furanose/furanoside/Pyranose/Pyranc b. Alfa/beta c. Reducing/ non-reducing
A: Pyranose-Saccharides with 6 membered ring with 5 carbons and 1 oxygen. A glycoside having pyranose…
Q: In an aqueous solution, d-glucose exists in equilibrium with two six-membered ring compounds. a.Draw…
A: Glucose is an monosaccharide and its formula is C6H12O6. Glucose contains 6 carbon atom, 12 hydrogen…
Q: 5.6. In the reaction mechanism of N-methylpropanamide reacting with LIAIH4 and followed by H20…
A:
Q: CHO 1. HCN HO- 2. Hа/Pd но 3. H30* CH2OH +
A: The reaction where aldose is treated with HCN and H2/Pd is known as Killiani-Fischer synthesis. Here…
Q: The carbonyl group on D-glucose can be reduced to d-glucitol. Identify the reagents that can chrry…
A: Glucose is a simple carbohydrate with one aldehydic and five Alcoholic functional groups. It's…
Q: 1. Draw the structure of the enol or enolate formed under the provided conditions, and estimate Keg…
A:
Q: Treatment with NaBH 4 converts aldose U into an optically inactive (meso) alditol V. Ruff…
A: Aldose: These are simple sugar or monosaccharide with carbon chain having Aldehyde group at top most…
Q: Draw the coupling product formed when each attached pair of compounds is treated with Pd(OAc)2,…
A: The coupling product formed when the given pair of compounds is treated with Pd(OAc)2, P (o-tolyl)3…
Q: 1. Draw the structure of the enol or enolate formed under the provided conditions, and estimate Keg…
A: Enol enolate formation
Q: Vhen the halohydrin is treated with NaH, a product of molecular formula C¸H§O is formed. Draw the…
A: When halohydrin is treated with NaH, epoxide is formed.
Q: Give a concise svnthesis for the molecules if they start from a benzene. a. F- -Br b. Но. OH C.
A:
Q: V. Give the product of the following SN1 reaction: CH3 H,O H3CH2C+E ČH,CH2CH3 to -Br a. Assign the…
A: V. Product of the given reaction,
Q: GMU 737664 What is the expected product A for the following intramolecular aldol reaction? O CH3 O…
A: In this question we have to tell the product of the reaction.
Q: What sugar units make up the structure shown? CH„OSO;- НО H H ÇOO- H NHCCH3 H H. K ÖH H он O…
A:
Q: H2SO4 + ОН HO a. A strong acid is used to catalyze this Fischer Esterification of acetic acid. What…
A:
Q: 6. the product of no.5 undergoes dehydration 7. D-arabinose reacts with dilute nitric acid with…
A: According to our guidelines I am answering first three questions only 6.The product of no.5…
Q: 20. The reaction of a pyranose to form a glycoside is shown below. The first step involves…
A: the solution is as follows:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Choose the product that is expected when the β-pyranose form of compound A is treated with excess ethyl iodide in the presence of silver oxide. The following information can be used to determine the identity of compound A: 1. The molecular formula of compound A is C6H12O6.2. Compound A is a reducing sugar.3. When compound A is subjected to a Wohl degradation two times sequentially, D-erythrose is obtained.4. Compound A is epimeric with D-glucose at C3.5. The configuration at C2 is R.trehalose is a disacharide that can be obtained from fungi sea uchins and insects. acid hydrolysis of trehalose yields only D-glucose. trehalose is hydrolysed by a-glucosidase but not b-glucosidase.methylation of trhalose followed by hydrolysis yield two molar equivalents of 2-3-4-6 -tetra-O-methyl-D-glucopyranose. deduce the structure of the trehalose using the experimental dataNaturally occurring compounds called cyanogenic glycosides, such as lotaustralin, release hydrogen cyanide, HCN, when treated with aqueous acid. The reaction occurs by hydrolysis of the acetal linkage to form a cyanohydrin, which then expels HCN and gives a carbonyl compound. (a) Show the mechanism of the acetal hydrolysis and the structure of the cyanohydrin that results. (b) Propose a mechanism for the loss of HCN, and show the structure of the carbonyl compound that forms.
- Ozonolysis, osmylation, and epoxide formation with mCPBA are all what type of reactions? a.) reduction b.)oxidation c) bothd) neitherDraw the product that is expected when the β-pyranose form of compound A is treated with excess ethyl iodide in the presence of silver oxide. The following information can be used to determine the identity of compound A: The molecular formula of compound A is C6H12O6. Compound A is reducing sugar. When compound A is subjected to a Wohl degradation two times sequentially, Derythrose is obtained. Compound A is epimeric with D-glucose at C3. The configuration at C2 is R.EXPLAIN IN DETAIL.What is the NAME of the substrate product when benzene is first treated with HNO3 and H2SO4, and THEN treated (in a second step) with Br2, and FeCl3 (or FeBr3)?
- When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw the structure of the product and indicateits stereochemistry.What product is formed when the esters in ginkgolide B, the chapter-opening molecule, are hydrolyzed in aqueous acid? Indicate the stereochemistry of all stereogenic centers.Indicate the product obtained by reacting alpha-D-Gulopyranose with NaBH4 in CH3OH.
- Draw a mechanism of the reduction of 188Re-perrhenate (Re(VII)) to Re(V) by using SnCl2.Esters with only one a hydrogen generally give poor yields in the Claisen condensation.Propose a mechanism for the Claisen condensation of ethyl isobutyrate, and explain whya poor yield is obtained.An optically active D-aldopentose (A) produced an optically inactive alditol (B) upon treatment with H2/Pt. When the aldopentose (A) was subjected to a Ruff degradation, D-aldotetrose (C) was generated. The aldotetrose (C) gave an optically active aldaric acid (D) upon oxidation with HNO3. D-aldopentose (A) can be prepared from D-threose by a Killani Fischer synthesis. Propose structure of A through D.



