Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter37: Dibenzalacetone By The Aldol Condensation
Section: Chapter Questions
Problem 10Q
Related questions
Question
use handwrited or present it with orderly manner when encoded please
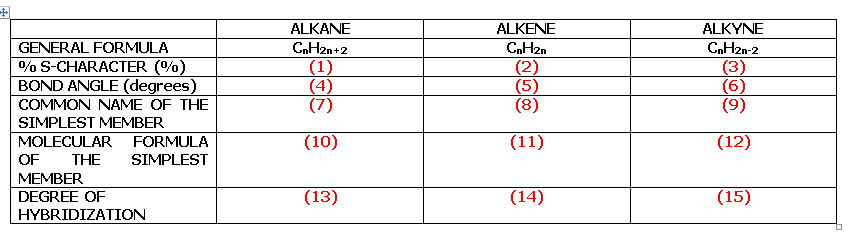
Transcribed Image Text:ALKANE
ALKENE
ALKYNE
GENERAL FORMULA
CnH2n+2
CnH2n
0% S-CHARACTER (%)
BOND ANGLE (degrees)
COMMON NAME OF THE
(1)
(4)
(7)
(2)
(5)
(8)
CnH2n-2
(3)
(6)
(9)
SIMPLEST MEMBER
MOLECULAR
FORMULA
(10)
(11)
(12)
OF
THE
SIMPLEST
MEMBER
DEGREE OF
(13)
(14)
(15)
HYBRIDIZATION
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning