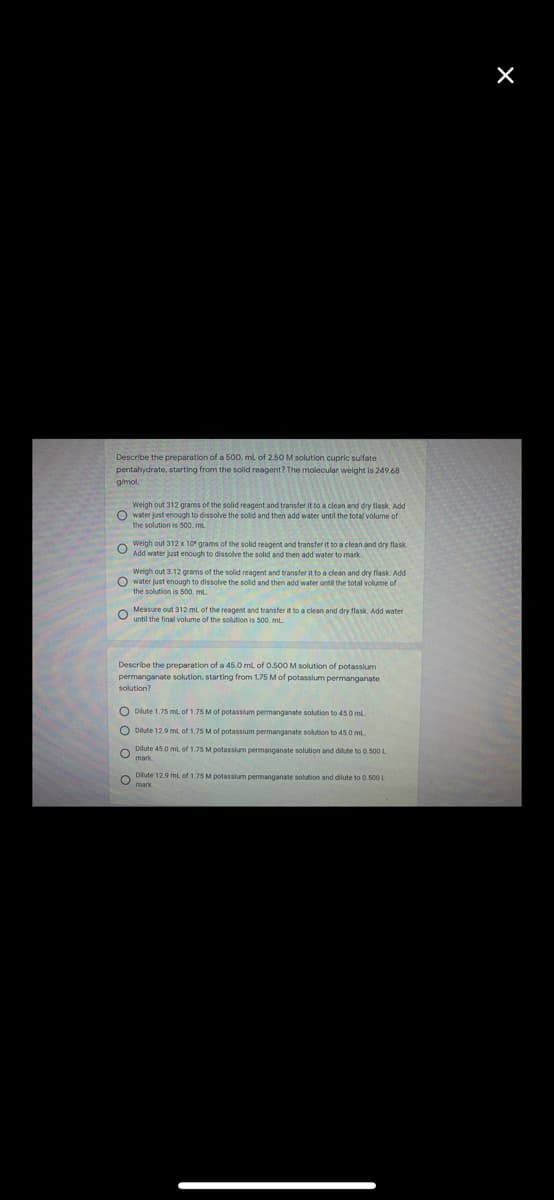
Describe the preparation of a 500. ml of 2.50 M solution cupric sulfate pentahydrate, starting from the solid reagent? The molecular weight is 249.68 gmol. Weigh out 312 grams of the solid reagent and transfer it to a clean and dry flask. Add O water just enough to dissolve the solid and then add water until the total volume of the solution is 500. mL Weigh out 312 x 1o" grams of the solid reagent and transfer it to a clean and dry flask. Add water just enough to dissolve the solid and then add water to mark Weigh out 3.12 grams of the solid reagent and transfer it to a clean and dry flask. Add O water just enough to dissolve the sold and then add water until the total volume of the solution is 500. ml. Measure out 312 ml of the reagent and transfer it to a clean and dry flask. Add water until the final volume of the solution is 500. mL Describe the preparation of a 45.0 ml of 0.500 M solution of potassium permanganate solution, starting from 1.75 Mof potasslum permanganate solution? O Dilute 1.75 ml of 1.75 M of potassium permanganate solution to 45.0 mL. Dilute 12.9 ml. of 1.75 M of potassium permanganate solution to 45.0 ml Dilute 45.0 ml of 1.75 M potassium permanganate solution and dilute to 0.500L mark Dilute 12.9 mt, of 1.75 M potassium permanganate solution and dilute to 0.500 L mark.
Describe the preparation of a 500. ml of 2.50 M solution cupric sulfate pentahydrate, starting from the solid reagent? The molecular weight is 249.68 gmol. Weigh out 312 grams of the solid reagent and transfer it to a clean and dry flask. Add O water just enough to dissolve the solid and then add water until the total volume of the solution is 500. mL Weigh out 312 x 1o" grams of the solid reagent and transfer it to a clean and dry flask. Add water just enough to dissolve the solid and then add water to mark Weigh out 3.12 grams of the solid reagent and transfer it to a clean and dry flask. Add O water just enough to dissolve the sold and then add water until the total volume of the solution is 500. ml. Measure out 312 ml of the reagent and transfer it to a clean and dry flask. Add water until the final volume of the solution is 500. mL Describe the preparation of a 45.0 ml of 0.500 M solution of potassium permanganate solution, starting from 1.75 Mof potasslum permanganate solution? O Dilute 1.75 ml of 1.75 M of potassium permanganate solution to 45.0 mL. Dilute 12.9 ml. of 1.75 M of potassium permanganate solution to 45.0 ml Dilute 45.0 ml of 1.75 M potassium permanganate solution and dilute to 0.500L mark Dilute 12.9 mt, of 1.75 M potassium permanganate solution and dilute to 0.500 L mark.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 50P
Related questions
Question
Transcribed Image Text:Describe the preparation of a 500. ml of 2.50 M solution cupric sulfate
pentahydrate, starting from the solid reagent? The molecular weight is 249.68
g/mol.
Weigh out 312 grams of the solid reagent and transfer it to a clean and dry flask, Add
O water just enough to dissolve the solid and then add water until the total volume of
the solution is 500. m
O Weigh out 312 x 10 grams of the solid reagent and transfer it to a clean and dry flask
Add water just enough to dissolve the solid and then add water to mark.
Weigh out 3.12 grams of the solid reagent
O water just enough to dissolve the solid and then add water until the total volume of
the solution is 500, ml
d transfer
oa clean and dry flask Add
Measure out 312 ml of the reagent and transfer it to a clean and dry flask. Add water
O until the final volume of the solution is 500. mL
Describe the preparation of a 45.0 ml of 0.500 M solution of potassium
permanganate solution, starting from 1.75 Mof potassium permanganate
solution?
O Dilute 1.75 ml of 1.75 M of potassium permanganate solution to 45.0 mL.
O Dilute 12.9 ml, of 1.75 M of potassium permanganate solution to 45.0 ml
Dilute 45.0 mt, of 1.75 M potassium permanganate solution and dilute to 0.500 L
mark
Dilute 12.9 ml of 1.75 M potassium permanganate solution and dilute to 0.500 L
mark.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning