describing the position of the particle, then f (t) is the instantaneous velocity of that particle. Find the instantaneous velocity in meters per second of a particle whose function is f (t) - 2t2-8t+3, when t- 5. f' (5) -
describing the position of the particle, then f (t) is the instantaneous velocity of that particle. Find the instantaneous velocity in meters per second of a particle whose function is f (t) - 2t2-8t+3, when t- 5. f' (5) -
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter9: Pre-quantum Mechanics
Section: Chapter Questions
Problem 9.4E
Related questions
Question
100%
Transcribed Image Text:M Gmail
YouTube
O Maps
GE News
G Google
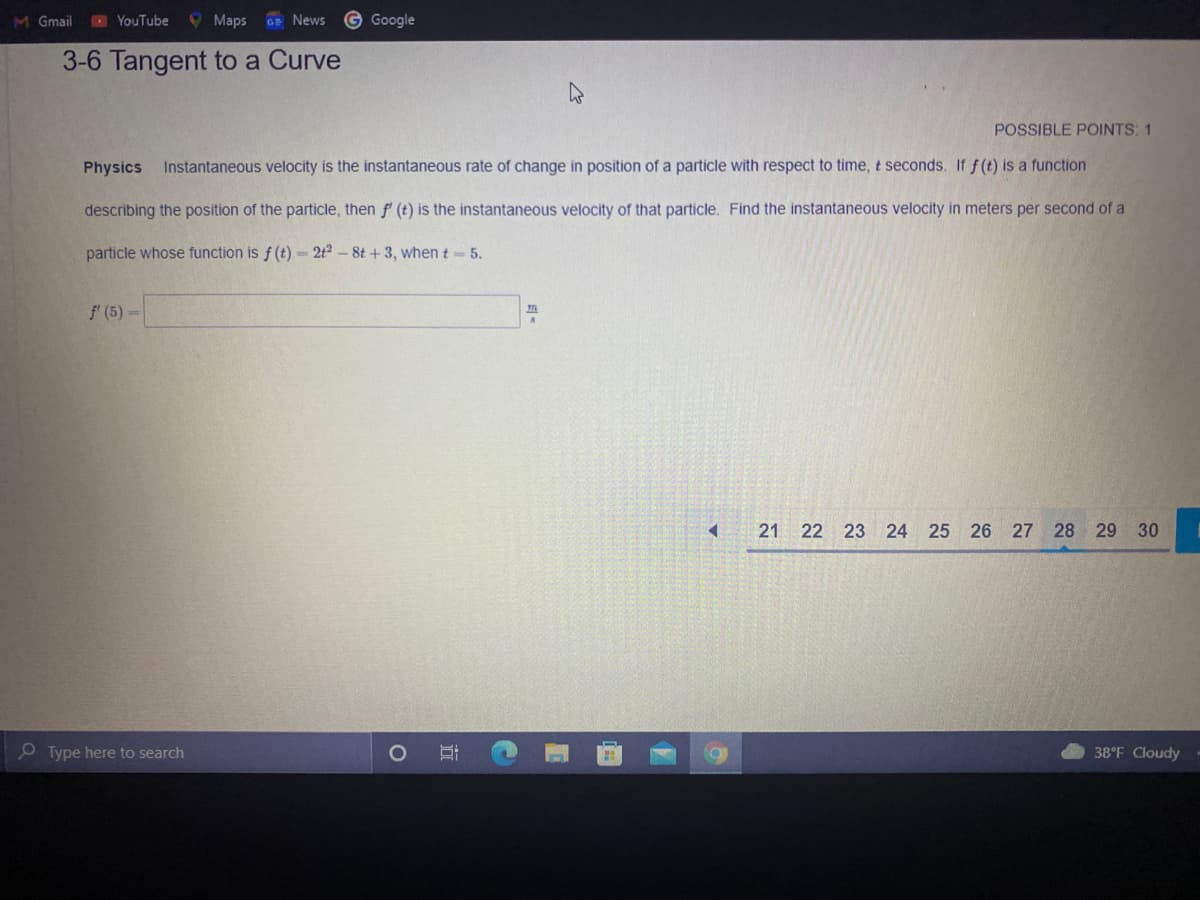
3-6 Tangent to a Curve
POSSIBLE POINTS: 1
Physics
Instantaneous velocity is the instantaneous rate of change in position of a particle with respect to time, t seconds. If f (t) is a function
describing the position of the particle, then f (t) is the instantaneous velocity of that particle. Find the instantaneous velocity in meters per second of a
particle whose function is f (t) = 2t2 – 8t + 3, when t 5.
f' (5)=
21 22 23 24 25 26 27 28 29 30
O Type here to search
38°F Cloudy
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning