Determine if a homogeneous solution forms when each pair of substances is mixed. Check all that apply. O CH3 CH2 CH2CH2CH3 and CH3 CH2 CH2CH2CH2CH3 O CBr4 and H2O O Cl2 and H2 0 Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining You have identified at least one incorrect pair of substances. Classify each compound as pola that "like dissolves like." For example, polar compounds dissolve homogeneously with other p
Determine if a homogeneous solution forms when each pair of substances is mixed. Check all that apply. O CH3 CH2 CH2CH2CH3 and CH3 CH2 CH2CH2CH2CH3 O CBr4 and H2O O Cl2 and H2 0 Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining You have identified at least one incorrect pair of substances. Classify each compound as pola that "like dissolves like." For example, polar compounds dissolve homogeneously with other p
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter3: Melting Points And Boiling Points
Section: Chapter Questions
Problem 8Q
Related questions
Question
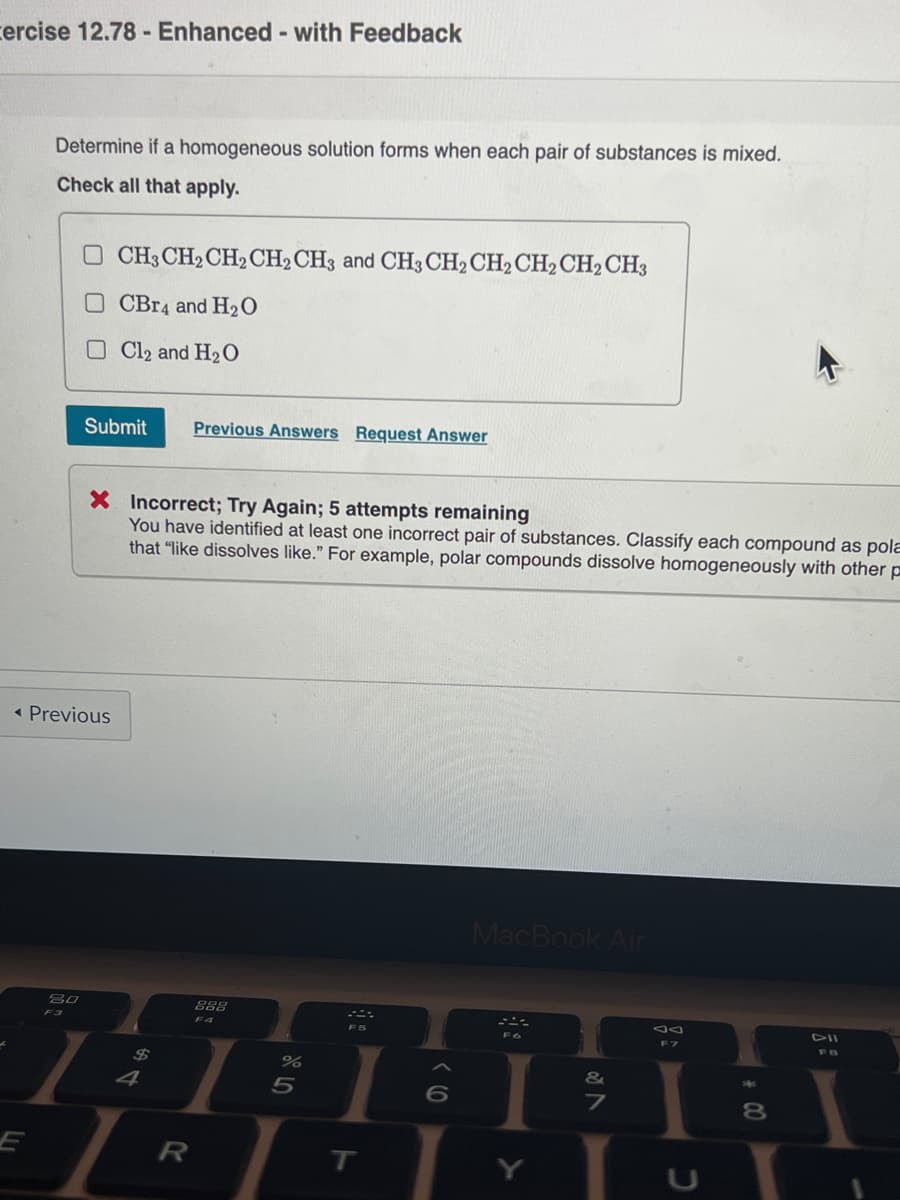
Transcribed Image Text:cercise 12.78- Enhanced- with Feedback
Determine if a homogeneous solution forms when each pair of substances is mixed.
Check all that apply.
O CH3 CH2 CH2CH2CH3 and CH3CH2CH2CH2 CH2CH3
O CBr4 and H2O
Cl2 and H2O
Submit
Previous Answers Request Answer
X Incorrect; Try Again; 5 attempts remaining
You have identified at least one incorrect pair of substances. Classify each compound as pola
that "like dissolves like." For example, polar compounds dissolve homogeneously with other p
« Previous
MacBook Air
80
888
F3
F4
%24
&
4
7
80
T
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole