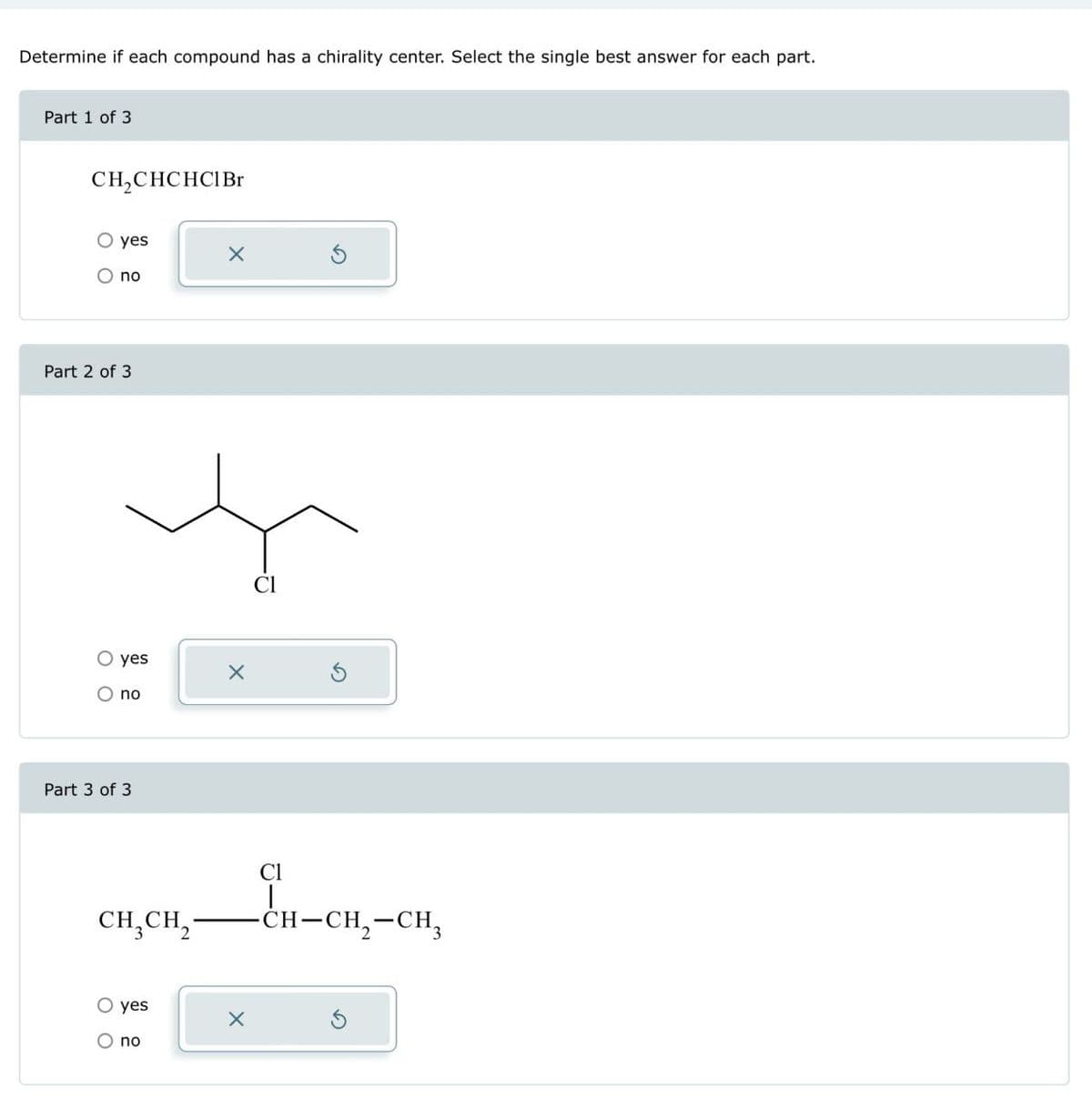
Determine if each compound has a chirality center. Select the single best answer for each part. Part 1 of 3. CH2CHCHCI Br ○ yes no Part 2 of 3 ○ yes O no Part 3 of 3 Cl CH3CH2 Cl | O yes O no -CH-CH2-CH3
Q: Please answer this question with steps thanks.
A:
Q: Liquid octane (CH 3 (CH 2 ) 6 CH 3 ) reacts with gaseous oxygen gas (O_{2}) to produce gaseous…
A: The objective of this question is to calculate the theoretical yield of water from the reaction of…
Q: Consider the reactionI2(g) + Cl2(g)2ICl(g)Using the standard thermodynamic data in the tables linked…
A: Step 1: Step 2: Step 3: Step 4:
Q: please help answer this question
A: Step :
Q: When the Pb2+ concentration is 5.00\times 10-4 M, the observed cell potential at 298K for an…
A: Step 1:Introduction to data :Given ,Concentration of Pb2+= 5.00 X 10-4Objective :Determine the…
Q: anode of an electrolytic cell must be connected to which terminal, positive or negative, of the…
A: The objective of the question is to understand the connection of anode in an electrolytic cell to…
Q: Draw the starting alkyl chloride that would produce this alkyne under these conditions. Drawing 1.…
A: Step 1: Step 2: Step 3:E2 elimination It typically requires a strong base to abstract the proton and…
Q: Conjugate acid base pKa 2) Consider the reaction B: () + H3O+ →>> BH(+) + H2O. For the following…
A: Note: The pKa values are not exactly correct there may be minor differences.
Q: I need answer expert solutions Need answer step by step
A:
Q: H2(g) + Cl2 (9) → 2HCl(g) 2NH3(g) → N2 (9) + 3H2 (9) CH4 (9) 202(g) → CO2(g) + 2H2O(g) AS 0 AS <0…
A:
Q: What would be the product of this reaction? BTBAC NaO. Br "CH₂ + H₂C CH₂Cl₂ A B
A: Step 1: Step 2: Step 3: Step 4:
Q: Which of the following will not undergo catalytic reduction in the presence of H, and metal…
A: The objective of the question is to identify which of the given groups, OH or CEN, will not undergo…
Q: 29. For the following reaction, determine the products and explain how you decided between a…
A: In deciding between elimination and substitution, several factors are considered:The nature of the…
Q: An analytical chemist is titrating 247.8 mL of a 1.100M solution of acetic acid (HCH, CO₂) with a…
A:
Q: For silicon, Si, the heat of fusion at its normal melting point of 1410 °C is 46.4 kJ/mol. The…
A:
Q: For a molecule, the phosphorescence lifetime without quenching is 1.0 s while with a quencher…
A: Given:Phosphorescence lifetime without quenching (τ0) = 1.0 sPhosphorescence lifetime with quencher…
Q: Sodium and water react according to the equation 2Na(s)+ 2H2O(l) → 2NaOH(aq) + H2(g) What number of…
A: Thank you.
Q: A mixture of NaHCO3 and Na2CO3 has a mass of 2.52 g. When treated with HCl(aq). 661 mL of CO2 gas is…
A: Given:…
Q: Zinc reacts with hydrochloric acid according to the reaction equation Zn(s) + 2HCl(aq) → ZnCl2(aq) +…
A: The objective of this question is to determine the volume of 2.50 M HCl(aq) required to react…
Q: Calculate the volume in of a 0.606M copper(II) sulfate solution that contains 500. mmol of copper 2…
A: The objective of this question is to calculate the volume of a 0.606M copper(II) sulfate solution…
Q: For the next compounds/ions, draw two (2) appropriate Lewis structures. One structure must obey the…
A: Lewis structures represent atoms by showing how they share electrons when they form a molecule.…
Q: Starting with the following alternative definition of Cp, and C₁: C = (S/S), C₁ = T(ƏS/ƏT)v show…
A: The problem asks us to show that Cp − Cv can be expressed as a function of PVT properties only,…
Q: Cengage Learning OWLv2 | Online teaching and C…
A: The objective of this question is to calculate the entropy change for the system when 2.46 moles of…
Q: L A Moving to another question will save this response. Question 6 For collisions to be successful,…
A: The question is asking about the conditions necessary for a successful collision between reactant…
Q: (3 points) Using the dissociation constant(ka), calculate the equilibrium concentrations of Cd2+ and…
A: The complex ion Cd(CN)42- dissociates into its component ions when dissolved in aqueous solution:…
Q: Please don't use hend raiting and step by step solutions
A: Acid catalysed Aldol Condensation :First enol forms enol by the protantion Enol reacts with…
Q: The structure of the amino acid tyrosine is shown below in its completely protonated form. PK 16 OH…
A: The question is asking for the structure of the amino acid tyrosine in its completely protonated…
Q: Chemistry Question
A: Here's a step-by-step guide on how to make chapati and dal:Chapati:Prepare the Dough:Take whole…
Q: A buffer is prepared by adding 5.00 g sodium acetate (MW = 82.0343 g/mol) and 10.0 mL of 5.00 %…
A: The objective of this question is to determine the amount of strong base that can be added to a…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the mechanism of the transformation from 3 to 4 .
A:
Q: A solution of hexane and heptane at 30 C with hexane mole fraction of 0.305 has a vapor pressure of…
A: The objective of the question is to find the vapor pressures of pure hexane and heptane at 30…
Q: Show the mechanism and all products formed when benzoic anhydride is treated with the nucleophile…
A: The objective of the question is to understand the reaction mechanism and identify the products when…
Q: 1-bromo-5-chloropentane LDA 1 eq a) Hg(OAc)2 b) NaBH NaOH (1 eq) CHOO Note this product has a ring.…
A: Step 1:a) In this reaction, • LDA is the strong base which will abstract most acidic proton to…
Q: Attempts Force Completion This test can be saved and resumed later. Your answers are saved…
A: The question is asking us to determine the direction of the shift in equilibrium when a base (NaOH)…
Q: 2. Show how you could synthesize each of the following molecules using a malonic ester synthesis…
A: Step 1: Step 2: Step 3: Step 4:
Q: A Suppose the molecule below is in acidic aqueous solution. Is keto-enol tautomerization possible? •…
A:
Q: identify the instructor - prepared maltrose as a - D - maltrose or B - D - maltrose. Describe the…
A: Maltose (malt sugar) is a disaccharide formed from two units of glucose joined with an α(1→4) bond.…
Q: help 12
A: Step 1: The hydroxide ion (OH-) from LiOH attacks the carbonyl carbon atom of the starting ester…
Q: Determine the enthalpy of combustion of liquid octane ( C8H18 ) at 25°C and 1 atm. Assume that the…
A:
Q: The chemical formulae of some acids are listed in the first column of the table below, and in the…
A: 1.) HClO (Weak Acid): A weak acid such as hypochlorous acid (HClO) only partially dissociates into…
Q: Predicting qualitatively how entropy changes with mixing and separation 1/3 For each system listed…
A: Step 1: Step 2: Step 3: Step 4:
Q: This is a multi- answer question. Please type your properly rounded answers in the next three…
A: reference value of Ka1 and Ka2 of H2SO3:Ka1= 1.7 x10-2 Ka2= 6.4x10-8 Step 1: We need to find the Kb…
Q: Consider an ideal gas enclosed in a 1.00 L container at an internal pressure of 24.0 atm. Calculate…
A: Step 1: first we have to find work done by the gas against external force.- pexternal(v2-v1). Step…
Q: None
A:
Q: G Decide whether each of the molecules in the table below is stable, in the exact form in which it…
A: Step 1: Step 2: Step 3: Step 4:
Q: 14 Moving to another question will save this response. Question 3 Consider the following reaction:…
A: Step 1: Step 2: Step 3: Step 4:
Q: The structure of the amino acid tyrosine is shown below in its completely protonated form. PK 16 OH…
A: The question is asking about the protonated form of the amino acid tyrosine and its pK values. The…
Q: You have a harmonic oscillator of mass 1.73 x 10-27 kg. There is an energy difference in adjacent…
A:
Q: Create a titration curve for each trail of pH vs. volume of base added. calculate the volume of NaOH…
A: The objective of the question is to analyze the titration data of an unknown acid with a standard…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Construct a model of CH3Br using MolView and answer the following: 1. Can it be superimposed on its mirror image? (Y/N) 2. Does CH3Cl contain a plane of symmetry? (Y/N) 3. Is CH3Cl chiral? (Y/N)For molecule CH3CH2CH(OH)CH3 I. Identify chiral centre II. Draw a pair of enantiomers using 3-dimensional structureThe following compound has only one asymmetric center. Why then does it have four stereoisomers? CH3CH2CH-BrCH2CH=CHCH3
- Consider CH3-CH(OH)-CH(OH)(Br). a.How many stereogeniccenters are in the molecule? b.How many stereoisomers are there for the compound? c.Draw the Fischer projection for each of the stereoisomer. Label each using I, II, etc. d.Which pairs are enantiomers? Which are diastereomers? e.Determine the absolute configuration of each chiral center in one pair of diastereomer.Construct a model of methane (CH4) using MolView and answer the following: Can it be superimposed on its mirror image? (Y/N) Does methane contain a plane of symmetry? (Y/N) Is methane chiral? (Y/N)a. How many asymmetric centers does the following compound have? b. How many stereocenters does it have? CH3CH-ClCH CHCH3
- Chris wants to select a compound that cannot exhibit chirality. The stockroom has available the following compounds: 2,3- dibromobutane, 1,4-dibromobutane, methylcyclopentane Which compound(s) should Chris choose?Draw a 3-D structure of a molecule with a molecular formula of C4H9Cl containing one chiral center and draw its mirror image.A) Please indicate the following compounds as chiral or achiral. B) Find 3 chiral centers and label them R or S

