Determine qrxn in joules for each trial and then convert to kJ. Pay careful attention to sign. What we no: Trail 1 -Qsoln=5013.22J Trail 2- Qsoln=5998.4J Trail3- Qsoln=5469.93J specific heat=4.02 Solve for qrxn not qsol.
Determine qrxn in joules for each trial and then convert to kJ. Pay careful attention to sign. What we no: Trail 1 -Qsoln=5013.22J Trail 2- Qsoln=5998.4J Trail3- Qsoln=5469.93J specific heat=4.02 Solve for qrxn not qsol.
Chapter30: Kinetic Methods Of Analysis
Section: Chapter Questions
Problem 30.2QAP
Related questions
Question
Determine qrxn in joules for each trial and then convert to kJ. Pay careful attention to sign.
What we no:
Trail 1 -Qsoln=5013.22J
Trail 2- Qsoln=5998.4J
Trail3- Qsoln=5469.93J
specific heat=4.02
Solve for qrxn not qsol.
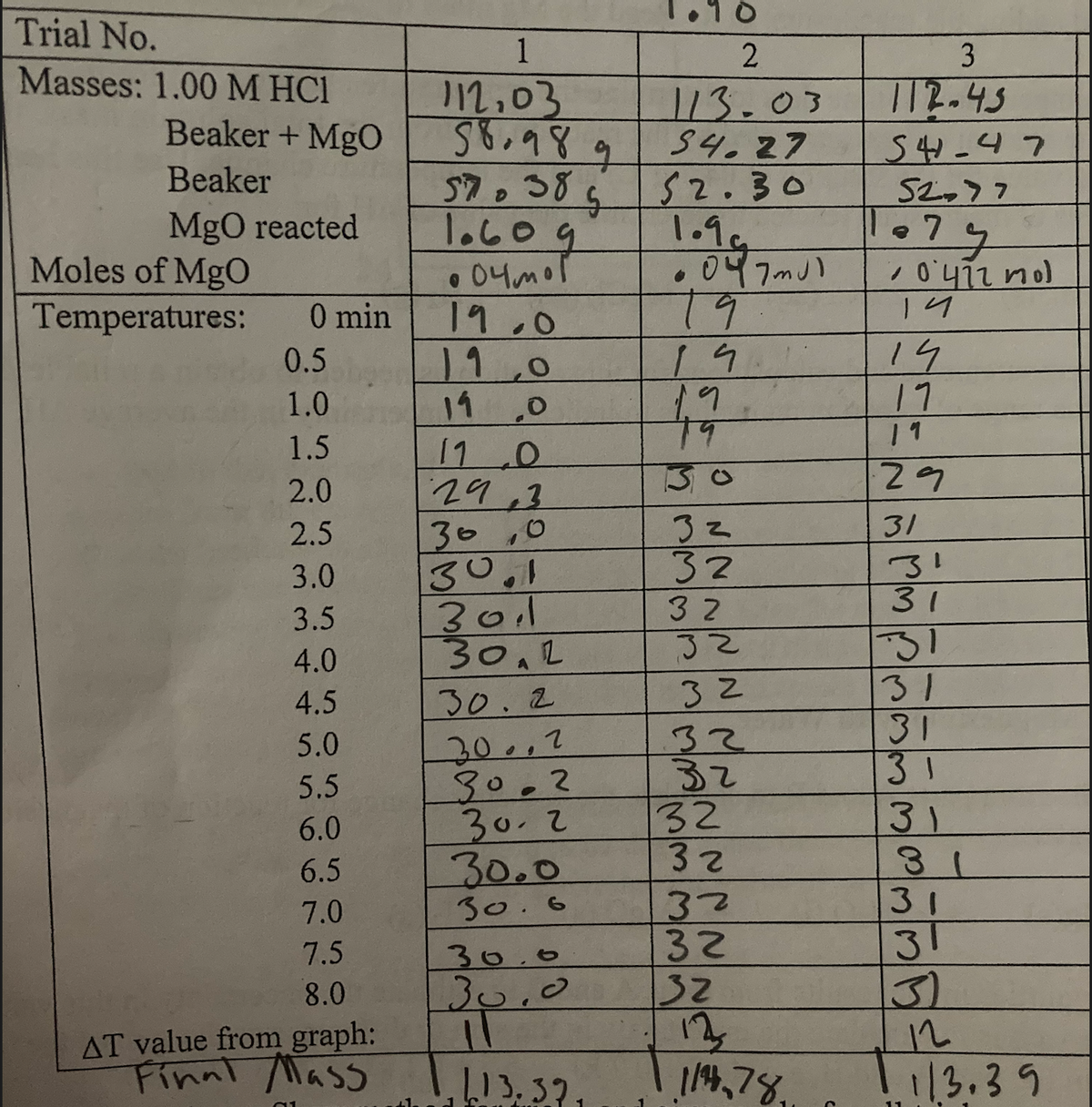
Transcribed Image Text:Trial No.
1
2
3
Masses: 1.00M HC1
Beaker+ MgO
112-43
12,03
S8.989
113.03
S4.27
52.30
1.9
• 047mJ)
19
544-47
S2.77
Beaker
1.60g
04mor
19.0
11,0
11.0
110
29,3
30,0
30.1
3011
30.2
MgO reacted
79
Moles of MgO
Temperatures:
0 min
14
14
17
1 1
0.5
19
1.0
6.
1.5
2.0
32
32
2.5
31
1.
3.0
32
31
3.5
4.0
31
31
32
30.2
30..2
30.2
30.2
30.0
30.6
4.5
32
32
32
32
5.0
5.5
31
31
31
6.0
6.5
32
32
7.0
31
3)
12
13.39
7.5
30.0
8.0
30,0
AT value from graph:
Finnl Mass
12
14,78,
13, 32,
333
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

