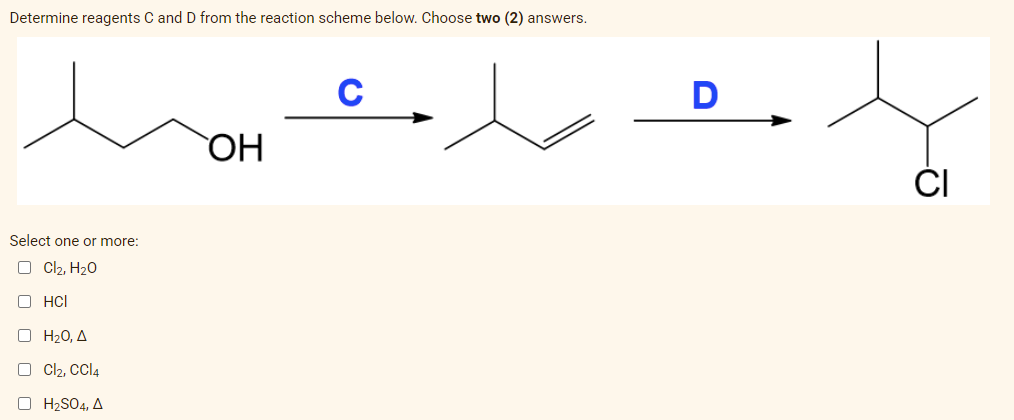
Determine reagents C and D from the reaction scheme below. Choose two (2) answers. Select one or more: O Cl₂, H₂O OHCI ο Η2Ο, Δ Cl₂, CCl4 H₂SO. A OH C D
Q: ssolve 14.7 grams of sodium hydroxide pellets in 100 ml of carbon-dioxide free water. ool the…
A:
Q: A chemist prepares a solution of potassium dichromate (K₂Cr₂O₂) by measuring out 9.94 μmol of…
A: A homogeneous mixture of two or more substances is known as a solution. The species which present in…
Q: Calculate the solubility, in mol/L, when lead (1) bromide (Ksp = 4.0 x 105) dissolves in an aqueous…
A:
Q: A 250.0 ml sample of gaseous hydrogen bromide, measured at 22.9°C and 0.930 atm, was dissolved in…
A:
Q: Questions and Problems Q1 Write the structure of the ionized form of phenylalanine # 10
A:
Q: 3. Calculate the pH of a buffer which is 0.2 mol/L with respect to ammonium sulphate and 0.1 mol/L…
A: Given -> Concentration of ammonium sulphate= 0.2 mole/L Concentration of NH3 = 0.1 mole/L Ka of…
Q: QUESTION 10 How many grams of MgO are produced when 40.0 grams of O2 react completely with Mg? 2Mg +…
A: Here we have to determine the mass of MgO produced when 40 g of O2 react completely with Mg in the…
Q: How many different proton environments are present in each of the following compounds? O A = 6; B=…
A:
Q: )? in kJ, round to the nearest whole number, and leave out units.
A:
Q: Determine whether the statement is true or false 1. The point at which an equivalent amount of the…
A: titration is chemical analysis in which the quantity of some constituent of a sample is determined…
Q: Identify the structure for the following H NMR spectrum OH 10 HO PPM
A: Please find your solution below : Spectroscopy is the study of interaction of radiation with matter.…
Q: What is the major organic product obtained from the following reaction? 01 2 SOCH, 1. NaOCH₂CH₂…
A: The given reaction is Michael addition followed by aldol condensation. This is also called Robinson…
Q: I ift ↑ Qlaccds X tab ΤΟΝ C caps lock H fn esc Re → O CHEMICAL REACTIONS Calculating molarity using…
A:
Q: Calculate the standard Gibbs free energy (in kJ/mol) of the reaction below. (Use R = 8.314 J/mol-K;…
A:
Q: Consider the electrochemical cell described by the following reaction at 310 K. The measured cell…
A: Cell potential is 0.242V , R=8.314J/mol-K, F= 96485C/mol , E° (reduction)= -0.17V, E°(oxidation)=…
Q: Write the standard base sequence of the messenger RNA that would cause a ribosome to make the…
A:
Q: hoose a reagent from the table to bring about the following conversion: Br If more than one step is…
A: The Answer to the following question is-
Q: If 50.0 mL of 0.435 M NaCl solution is diluted to 225.0 mL, what is the concentration of the diluted…
A: Given data is as follows: The initial volume of NaCl solution V1= 50.0 mL The initial concentration…
Q: Cuien H₂₂ (g) + F₂ (g) = 2HF (g) Find the partial pressure of HF? Crien An empty Steel Container is…
A:
Q: If 6.43 g of CuNO, is dissolved in water to make a 0.550 M solution, what is the volume of the…
A:
Q: Question 11 of 20 Which classification of lipid (s) has one or more fatty acids? A) Waxes B)…
A: Here we to predict which of the following lipids contains one or more fatty acids. The above…
Q: Part A For neutral molecules, which statements about covalent Lewis structures are true? Drag each…
A: For neutral molecules, the following statement is correct about the Lewis Structure:
Q: 5.48 Draw all possible configurational isomers of the molecule shown here. of the following species…
A: The IUPAC NAME FOR THE GIVEN COMPOUND IS., 2,3-dibromo-4,5-dichloroheptane
Q: Water heat is of 4.18j?g c° what is the quantity of energy in kJ required to increse the temperature…
A:
Q: Tarnish on iron is the compound FeOFeO. A tarnished iron plate is placed in an aluminum pan of…
A:
Q: How many mols of HBr are used to neutralize 18.66 mL of a 0.2299 M Ca(OH)₂ solution?
A:
Q: The phase diagram for argon is shown below: a) b) c) P atm 48 1.00 0.68 83.8 83.9 T Kelvin 87.4 What…
A:
Q: Provide IUPAC names for the following compounds. (a) OH (b)
A: We follow the below steps designed by IUPAC to assign IUPAC name to an organic molecule.
Q: Consider the electrochemical cell described by the following reaction at 310 K. The measured cell…
A:
Q: 5.53 For each of the following pairs, determine if the compounds have the same boiling point or…
A: Structure of compound with same molecular formula define the properties like boiling point will be…
Q: The pka of ethanoic acid, (CH3COOH) (0.1 M), is 2.6 at 25°C. Write the balanced chemical equation…
A:
Q: From mass to moles There are two unknowns in this problem - the grams of potassium carbonates and…
A: Let the mass of the mixture (Na2CO3 + K2CO3) = m Moles of total HNO3 used = n To find: Mass of…
Q: How many more times basic is something with a pH 13 as compared to a pH of 11?
A: Hydroxide ion concentration measure the basicity of the solution. If pH more then 7 then solution is…
Q: If 2.68 g of benzaldehyde are involved with the mixed aldol condensation reaction, how many mL of…
A:
Q: Considering intermolecular forces for what reason would nitrogen trichlorode have such a high…
A: Given, Considering intermolecular forces for what reason would nitrogen trichloride have such a…
Q: Classify the following reaction as an Sn1, Sn2, E1, E1cB, E2 reaction. Can you show me the leaving…
A:
Q: What is the IUPAC name of CH₂CH3 CH3CHCHCH₂CH₂CH₂CH₂CH3 3 CH3 O2-ethyl-3-methyloctane…
A:
Q: What is K for a reaction if ∆G° =-216.0 kJ/mol at 25°C (or 298 K)? (R = 8.314 J/mol ・ K)
A:
Q: For the equilibrium system below, determine which direction the equilibrium will shift under each…
A:
Q: How many grams of potassium chlorate are there At the completion of reactions?
A: Given that, the decomposition reaction of potassium chlorate, KClO3. We have to tell the amount of…
Q: Which one of the following structures is a free radical? a) NO+ b) SO+ c) CO d) ClO+
A: Free radical : Free radicals are those species which contains one unpaired electron. Example :…
Q: Problem 2. Draw a peptide bond between two amino acids: a) circle the carboxyl and amino group…
A:
Q: What is the correct balanced redox reaction for Al³+ (aq) + Ag(s) Al(s) + Ag* (aq) A1³+ (aq) +…
A:
Q: Nitriloacetic acid is an aminopolycarboxylic acid
A: (10) Potentiometric methods can be used for end-point detection in EDTA titration of metal ions for…
Q: The normal freezing point of water, H₂O is 0.00 °C and its Kip value is 1.86 °C/m. Assuming complete…
A: Colligative properties: The colligative properties are those properties of solutions that depend on…
Q: Using the partial equations below, determine the enthalpy of formation of nitric oxide (NO). Hint:…
A:
Q: Which of the following is aromatic? www www.
A: Aromatic compound which obey 4n+2 rule and it should be cyclic, conjugated and planar.
Q: How many mols of NaOH are present in 15.82 mL of a 0.2106 M NaOH solution?
A: The moles of NaOH that are present is = 0.00333 mol
Q: (UV spectroscopy lab, organic chemistry 2) Acetaldehyde shows two UV bands, one with a λmax of 289…
A: To assign the following bands to transition, we would use the electronic transition diagram in UV…
Q: 1. a) What is meant by the term "buffer solution"?
A: Any solution which can resist any change in pH on addition of small amount of acid or base called…
Step by step
Solved in 2 steps with 1 images

- A student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major products of formula C7H11Br. (a)Rank these three intermediates from most stable to least stable.Kk.174. Please fill out this reaction mechanism with the missing reagents and intermediate products with a complete arrow-pushing mechanism.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heat
- Reaction of HBr with 2-methylpropene yields 2-bromo-2-methylpropane. What is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction.3H. Put it all together. Predict the E1 products of the following reaction. Label alkenes as E/Z. 4. Predict the E1 products of the following reaction. Label akenes as E/Z.For each reaction, show the generation of the electrophile and predict the products.(a) benzene + cyclohexene + HF (b) tert@butyl alcohol + benzene + BF3
- Please give a detailed stepwise mechanism for the following reactions of Q.1 and Q.2. All arrows, charges, intermediates, and resonance structures must be shown.Draw the products, including their configurations, obtained from the reaction of 1-ethylcyclohexene with the following reagents:a. HBr b. H2, Pd/C c. R2BH/THF, followed by HO– , H2O2, H2O d. Br2/CH2Cl2For each reaction, show the generation of the electrophile and predict the products.(a) benzene + cyclohexene + HF (b) tert@butyl alcohol + benzene + BF3(c) tert@butylbenzene + 2@methylpropene + HF
- Propose a mechanism for each reaction, showing explicitly how the observed mixtures of products are formed. cyclopenta-1,3-diene + Br2 S 3,4-dibromocyclopent-1-ene + 3,5-dibromocyclopent-1-eneTreatment of cis-2-bromocyclohexanol with NaOH yields cyclohexanone instead of an epoxide. Draw curved arrows to show the movement of electrons in this step of the reaction mechanism.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acid

