Part A For neutral molecules, which statements about covalent Lewis structures are true? Drag each item to the appropriate bin. View Available Hint(s) True For a neutral molecule, the number of electrons in the Lewis structure is the sum of the valence electrons for the atoms. Hydrogen atoms are often the central atom of a Lewis structure. Reset Help Electrons of covalent compounds may be shared between atoms. Each atom of a Lewis structure must have eight electrons. False
Part A For neutral molecules, which statements about covalent Lewis structures are true? Drag each item to the appropriate bin. View Available Hint(s) True For a neutral molecule, the number of electrons in the Lewis structure is the sum of the valence electrons for the atoms. Hydrogen atoms are often the central atom of a Lewis structure. Reset Help Electrons of covalent compounds may be shared between atoms. Each atom of a Lewis structure must have eight electrons. False
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter2: Lewis Structures
Section: Chapter Questions
Problem 10CTQ
Related questions
Question
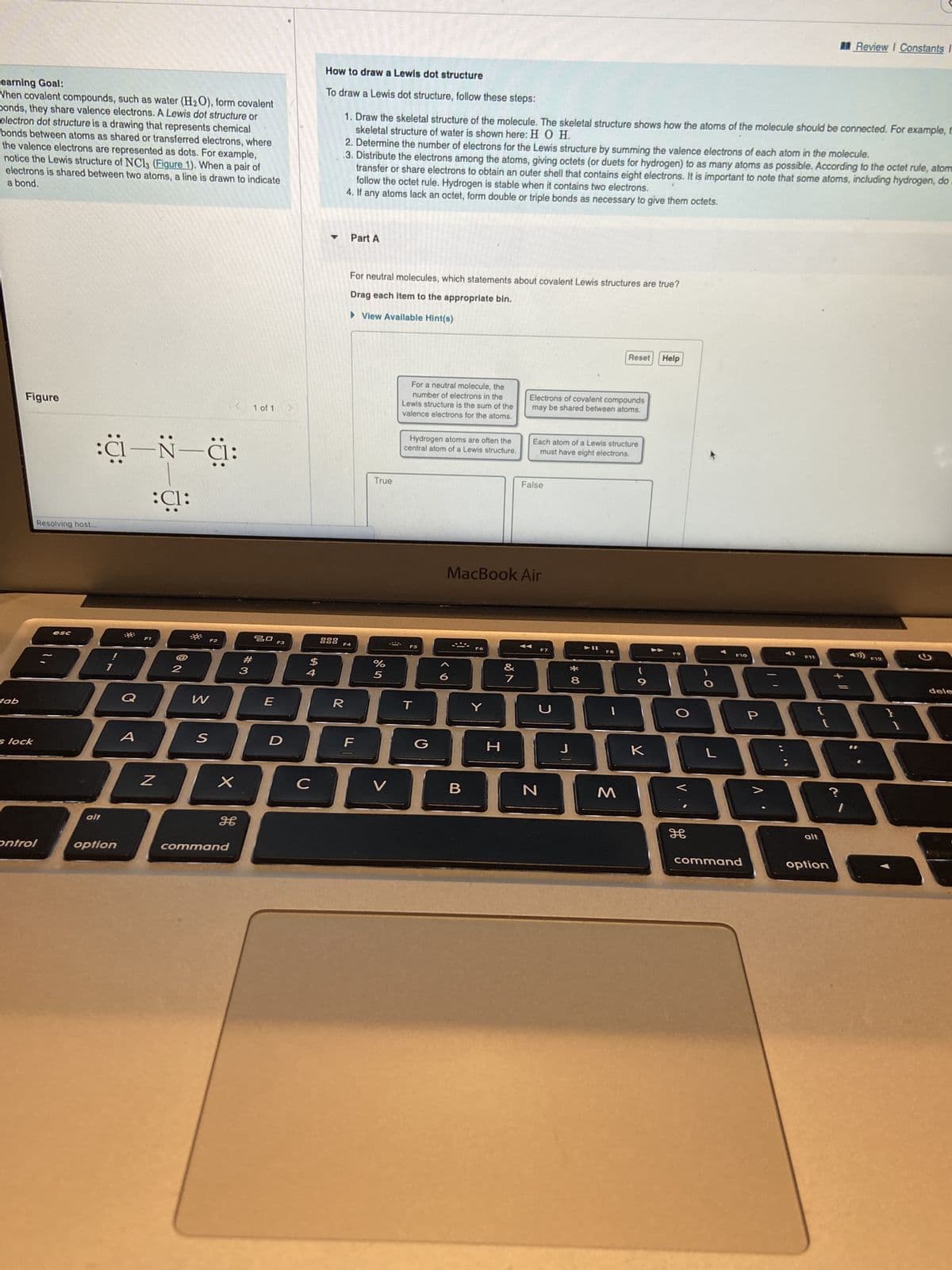
Transcribed Image Text:Learning Goal:
When covalent compounds, such as water (H₂O), form covalent
Donds, they share valence electrons. A Lewis dot structure or
electron dot structure is a drawing that represents chemical
bonds between atoms as shared or transferred electrons, where
the valence electrons are represented as dots. For example,
notice the Lewis structure of NC13 (Figure 1). When a pair of
a bond.
electrons is shared between two atoms, a line is drawn to indicate
tab
Figure
s lock
Resolving host...
:C—N—C:
:C1:
esc
control
alt
F1
option
Z
2
W
S
X
H
command
#
1 of 1
3
20 F3
$
10809009
4
E
D
C
How to draw a Lewis dot structure
To draw a Lewis dot structure, follow these steps:
skeletal structure of water is shown here: H O H.
1. Draw the skeletal structure of the molecule. The skeletal structure shows how the atoms of the molecule should be connected. For example, t
2. Determine the number of electrons for the Lewis structure by summing the valence electrons of each atom the molecule.
.3. Distribute the electrons among the atoms, giving octets (or duets for hydrogen) to as many atoms as possible. According to the octet rule, atom
transfer or share electrons to obtain an outer shell that contains eight electrons. It is important to note that some atoms, including hydrogen, do
follow the octet rule. Hydrogen is stable when it contains two electrons.
4. If any atoms lack an octet, form double or triple bonds as necessary to give them octets.
Part A
For neutral molecules, which statements about covalent Lewis structures are true?
Drag each item to the appropriate bin.
View Available Hint(s)
F4
R
True
F
%
5
For a neutral molecule, the
number of electrons in the
Lewis structure is the sum of the
valence electrons for the atoms.
V
Hydrogen atoms are often the
central atom of a Lewis structure.
F5
T
G
B
MacBook Air
Y
H
&
Electrons of covalent compounds
may be shared between atoms.
7
Each atom of a Lewis structure
must have eight electrons.
False
J
N
J
*00
Reset
8
M
к
Help
F9
V
L
F10
command
88
{
P
F11
. V
Review I Constants I
alt
option
F12
G
dele
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning