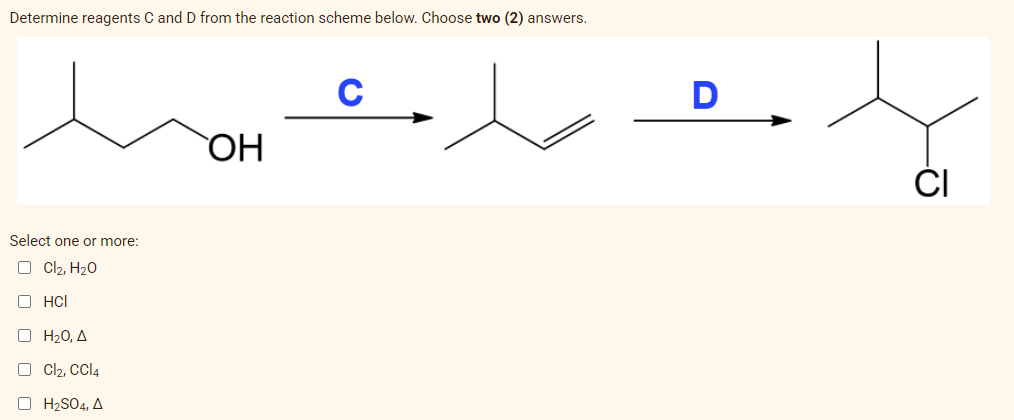
Determine reagents C and D from the reaction scheme below. Choose two (2) answers. Select one or more: Cl₂, H₂O ⒸHCI ο Η2Ο, Δ Cl₂, CCl4 OH₂SO4, A OH C D -Ō
Q: Wt of precipitate (Silver chloride) MW of silver nitrate MW of silver chloride Meg silver nitrate…
A:
Q: Ca(OH)2 (aq) + H₂SO4 (aq) →+ 2 H₂O (1) + CaSO4 (aq) (This is LIMITING REACTANT: H.SO, is the…
A: Above reaction is is Acid base reaction because salt and water are form as product.
Q: The pka of ethanoic acid, (CH3COOH) (0.1 M), is 2.6 at 25°C. Write the balanced chemical equation…
A:
Q: What diene and dienophile are used in the Diels-Alder route to the compound shown? O CO₂CH3 + A
A:
Q: Calculate the reaction temperature of the electrochemical cell described below which measures a…
A:
Q: A 0.4320 -g sample of pure iron (55.847 g/mol) wire was dissolved in acid, reduced to the +2 state,…
A:
Q: What is the amount of excess reactant left over at the end of the reaction. Use the first image as…
A: Since H2SO4 produces the smallest amount of the products, it is the limiting reactant. Hence we…
Q: I ift ↑ Qlaccds X tab ΤΟΝ C caps lock H fn esc Re → O CHEMICAL REACTIONS Calculating molarity using…
A:
Q: One of the molecular orbitals of the formyl cation, HCO+ (H-C=O)*, shown below. Identify the type of…
A: Hydrogen has 1s orbital , so it can bind with another through sigma bonding interaction
Q: 2. a) Calculate the pH of 0.12 mol/L ethanoic acid (K₂ = 1.7 x 105 mol/L). b) Calculate the mass of…
A: Since the given question is a multipart question, hence I solved first three parts according to…
Q: How many grams of lithium nitrate will be needed to make 200.0 grams of lithium sulfate, assuming…
A: The balanced chemical equation is: Pb(SO4)2 + 4LiNO3 →Pb(NO3)4 + 2Li2SO4(s)
Q: Question 9 A 0.2463 -g sample of pure iron (55.847 g/mol) wire was dissolved in acid, reduced to the…
A:
Q: One of the steps in the production of sulfuric acid involves the catalytic oxidation of sulfur…
A: According to the question we have the reaction of catalytic oxidation of sulfur dioxide. 2SO2 (g) +…
Q: the solubility of silver sulfate in water is determined to be 570 mg/100mL based on the given…
A: A question based on solutions that is to be accomplished.
Q: 5.76 2,3-Dibromoprop-1-ene (C3H4Br₂) has four H atoms. Suppose that any of these H atoms can be…
A: Constitutional isomers :- The molecules which have same molecular formula but different…
Q: You see a flash of lightning and then hear the thunder 5.2 seconds later. If the temperature is 26…
A: Given, Temperature,T 26°C=(26+273)K=299 K
Q: Provide IUPAC names for the following compounds. (a) OH (b)
A: We follow the below steps designed by IUPAC to assign IUPAC name to an organic molecule.
Q: Propyne is reacted with NaNH2. The major organic product of this reaction is then reacted with…
A: We can write the IUPAC name of alkyne using following rules Identify parent chain ( chain with…
Q: Question 5 Calculate the percentage of MnO2 (MW 86.937 g/mol) in a mineral specimen if the 12…
A: We are given : Volume of Na2S2O3 used = 29.89 mL = 29.89 × 10-3 L Molarity of Na2S2O3 = 0.0814 M…
Q: Wt of precipitate (Silver chloride) MW of silver nitrate MW of silver chloride Meg silver nitrate…
A:
Q: Which of the following compounds will display two triplets and a singlet in the ¹H NMR spectrum? □…
A:
Q: A chemist prepares a solution of silver(II) oxide (AgO) by measuring out 0.0121 umol of silver(II)…
A: Molarity is defined as the moles of a solute per liters of a solution. Molarity is also known as the…
Q: Determine the enthalpy of reaction for the following: H2(g) + ¹/2O2(g) → H₂O(g)
A: Given reaction: H2(g) + 12 O2(g) → H2O(g) We have to find the enthalpy of reaction.
Q: A common demonstration in chemistry courses involves adding a tiny speck of manganese(IV) oxide to a…
A: In Decomposition reactions chemical species break up into simpler molecules. At very high…
Q: Predict the number of signals expected (disregarding splitting) in the 1H spectrum of CH3-OCH2CH3
A: Chemically non equivalent hydrogen atoms will have different chemical shift and therefore give rise…
Q: A gas sample containing 12.0 g of He has a volume of 14.0 L. What will the volume be if 1.6 g of H2…
A:
Q: Calculations for experimentally determining R Trial 1 Trial 2 Mass of Mg ribbon (g) 0.031 0.039…
A: Trial 1: Given that, Mass of magnesium ribbon =0.031 g Temperature of H2(g) = 24.0 °C Volume of…
Q: 89. Give the balanced equation for each of the following chemical reactions: a. Glucose (C,H,O,)…
A: Given,a. Glucose C6H12O6 reacts with oxygen gas b. Solid iron III sulfide reacts with gaseous…
Q: For the reaction: PC13(g) + Cl2(g) = PC15(g) at 85°C, Kp = 1.19 If one starts with 2.00 atm pressure…
A: Kp is equilibrium constant in terms of pressure and is equal to the ratio of the product of…
Q: 5.69 Carvone is present in many essential oils. It is chiral, and its two enantiomers are shown…
A: carvone is a chiral molecule; its enantiomers have different physical and chemical properties.The…
Q: A buffer prevents drastic changes in PH, meaning for this one it should be ranging somewhere +/-…
A: From #Q.6, the buffer solution has 0.5 mmol of conjugate base, sodium acetate(CH3COONa), and 0.4…
Q: Calculate the molar solubility of Ag2CrO4 in a solution that is 0.01 M in AgNO3. Ksp of Ag2 CrO4 =…
A:
Q: What is the molarity of a solution containing 2.75 moles of NaCl in 5.00 L of solution?
A: Definition of molarity- Molarity is the ratio of moles of solute to the volume of the solution.…
Q: Calculate the standard Gibbs free energy (in kJ/mol) of the reaction below. (Use R = 8.314 J/mol-K;…
A: Given reaction is : 6Cu (s) + Cr2O72- (aq) + 14H+ (aq) -------> 6Cu+ (aq) + 2Cr3+ (aq) + 7H2O…
Q: Calculate the equivalence point potential for the titration of 50.0 ml of 0.02 M Ce with 33.33 mi of…
A:
Q: What is the steroid nucleus? A) 3 cyclohexane rings and 1 cyclopentane ring fused together B) 2…
A: Asteroid: Asteroid is a class of lipid. Asteroid have large number of carbon and hydrogen which…
Q: Which of the following is the best description of methane clathrate hydrate? Select one: a. Methane…
A: When compounds makes bonds with crystal water molecules then hydrates of that compounds is formed.
Q: Suppose when Millikan did his experiment and measured the charges on various oil droplets, he got…
A: Millikan in his experiment examined the motion of single tiny drops of oil that picked up static…
Q: need help on the following with explanation Please do both correctly neat and clean otherwise don't…
A: (1) German chemist and physicist Erich Hückel proposed a theory to help determine if a planar ring…
Q: The product P in the following chemical reaction will be 0= CI 1. NaN3 2. Heat 3. H₂O P
A:
Q: 0.00188 g of AgCl dissolves in one liter of H2Oat 25 * c. Write the balanced chemical equation for…
A: 0.00188 g of AgCl is dissolved in 1 L of H2O at 25 °C7.8 × 10- 5 moles of silver chromate is…
Q: Part A For neutral molecules, which statements about covalent Lewis structures are true? Drag each…
A: For neutral molecules, the following statement is correct about the Lewis Structure:
Q: The phase diagram for argon is shown below: a) b) c) P atm 48 1.00 0.68 83.8 83.9 T Kelvin 87.4 What…
A:
Q: (UV spectroscopy lab, organic chem 2) Perhaps it is unsurprising that cyclohexane and ethanol are…
A: To examine a compound in UV spectroscopy, certain solvents are used. The solvent are chosen such…
Q: HO است به HO 5 4 3 OH The Haworth projection of D-altrose is shown. Which anomer is depicted?…
A: The carbon atom that initially contains the carbonyl functional group in open chain sugar and in the…
Q: Benzene was reacted with propanoyl chloride in the presence of a Lewis acid catalyst to form…
A: When alcohol is treated with acetic anhydride then ester and acetic acid is formed as shown :
Q: = 5. a) Calculate the pH of a buffer solution which is 0.1 mol/L with respect to HCN (Ka 4.9 x 10-10…
A: a). pH of a buffer solution can be calculated with the help of Henderson - Hasselbalch equation : pH…
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A: The polarity of a bond depends on electronegativity difference between the two atoms sharing the…
Q: 7. Which of the following is aromatic? Å
A: We need to find the aromatic compound among these.
Q: A voltaic cell consists of two half-cells. One of the half-cells contains a platinum electrode…
A: According to the question, we have the half cell reaction of the voltaic cell which is:- 2BrO3-…
Step by step
Solved in 3 steps with 3 images

- A student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major products of formula C7H11Br. (a)Rank these three intermediates from most stable to least stable.Kk.174. Please fill out this reaction mechanism with the missing reagents and intermediate products with a complete arrow-pushing mechanism.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heat
- Reaction of HBr with 2-methylpropene yields 2-bromo-2-methylpropane. What is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction.3H. Put it all together. Predict the E1 products of the following reaction. Label alkenes as E/Z. 4. Predict the E1 products of the following reaction. Label akenes as E/Z.For each reaction, show the generation of the electrophile and predict the products.(a) benzene + cyclohexene + HF (b) tert@butyl alcohol + benzene + BF3
- Please give a detailed stepwise mechanism for the following reactions of Q.1 and Q.2. All arrows, charges, intermediates, and resonance structures must be shown.Draw the products, including their configurations, obtained from the reaction of 1-ethylcyclohexene with the following reagents:a. HBr b. H2, Pd/C c. R2BH/THF, followed by HO– , H2O2, H2O d. Br2/CH2Cl2For each reaction, show the generation of the electrophile and predict the products.(a) benzene + cyclohexene + HF (b) tert@butyl alcohol + benzene + BF3(c) tert@butylbenzene + 2@methylpropene + HF
- Propose a mechanism for each reaction, showing explicitly how the observed mixtures of products are formed. cyclopenta-1,3-diene + Br2 S 3,4-dibromocyclopent-1-ene + 3,5-dibromocyclopent-1-eneTreatment of cis-2-bromocyclohexanol with NaOH yields cyclohexanone instead of an epoxide. Draw curved arrows to show the movement of electrons in this step of the reaction mechanism.Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acid

