Q: e The illustration to the left represents a mixture of nitrogen (blue) and hydrogen (light blue)…
A: All answers are uploaded Explanation:
Q: The compound below is classified as what type of compo OCH,0
A: Functional groups are atoms or a group of atoms that are characterized by distinctive chemical…
Q: Please provide explanations
A: For each alkene and reagent pair, you would need to consider the reaction mechanism involved. Here's…
Q: Show how you would make the TM from the given SM. HO HOC "COH
A: In the first step we have to reduce the keto group into alcohol group by the use of LiAlH4 /mild…
Q: Consider the reaction of CO2 and solid carbon described by the chemical reaction below. Determine…
A: Answer:For any reaction, value of equilibrium constant KC is equal to the ratio molar concentration…
Q: Q2. A) Determine the monomer unit of the following polymer CH3 CH3 CH3 CH₂-C-CH2-C-CH2-7 CO2CH3…
A: A monomer is a small molecule that can chemically bond to other monomers to form a polymer. Monomers…
Q: A. For each reaction 1) Provide the missing reagents/conditions or major organic products as…
A: The objective of this question is to identify appropriate reagents and conditions for specific…
Q: When you work on a modern SEM, how do you determine the charging voltage on the specimen surface?
A: A contemporary Scanning Electron Microscope (SEM) is a powerful piece of equipment that can image…
Q: Question 6 What are the correct configurations of the chiral centers in the following compound? 10=0…
A: The Cahn-Ingold-Prelog (CIP) priority rules, also known as the sequence rules, are used to determine…
Q: i need the answer quickly
A:
Q: Question 10: Which of the alkyl bromides listed would work as the alkyl halide in step 2 of the…
A: Since you have asked multipart-type questions, we will solve only the first three questions for you.…
Q: B. Choose the major product for reaction I C. Choose the correct reagent for reaction IIA. HBrB.…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: You have 50.0mL of 0.520M NH3. Kb = 1.8x10-5 You add 15.0mL of a 0.05M HCl solution. What is the pH…
A: The objective of this question is to calculate the pH of a solution after the addition of a certain…
Q: write a balanced equation for the decomposution reaction that occurred in experiment 2. Include…
A:
Q: < Solid Ca3(PO4)2 is placed into 10.0 L of water. When equilibrium is established, the concentration…
A: Equilibrium constant Kc for a reaction is defined as the product of the molar concentrations of the…
Q: For the following reactions, provide an organic product in each answer box. If only one box is…
A: In the presence of a weak nucleophile, substitution proceeds via the SN1 mechanism. An SN1 mechanism…
Q: Show how you would make the TM from the given SM. HO O HOC "COH
A: In the first step, we have to reduce the keto group into the alcohol group by the use of LiAlH4…
Q: 2. The major product of the following reaction is, KOH Br H H₂O
A: When a carbon with good leaving group is electron poor centre due to -I effect of leaving group then…
Q: In a titration experiment, 46.0 mL of 0.085 M acetic acid, CH3COOH, is titrated with the 0.250 M…
A: Axid-base titration is a useful technique for the determination of the unknown concentration with…
Q: What term best describes the relationship between the structures below? H, CI H₂N Enantiomers…
A: We have to choose the correct option.
Q: 7. Which of the following carbocations is least likely to undergo a 1,2-rearrangement? Bubble in…
A: Carbocation can be defined as the species in which a carbon atom carries positive charge. In the…
Q: Draw the major product of the given reaction. Include minimized formal charges. H NaOEt NO2 EtOH A…
A: The reaction used for formation of beta nitroalcohols from aldehydes and ketones using nitroalkanes…
Q: 7. Which bond is correctly labeled with a dipole arrow? + + C-H O-H N-H C-Cl C-C a. b. C. d. a.…
A: Find out the correctly labeled arrow.
Q: A diprotic acid, H2A, has acid dissociation constants of Kal = 3.78 x 104 and Ka2 = 4.21 x 10-11.…
A: A diprotic acid can dissociate to produce two H+ ions.
Q: While ethanol (CH3CH2OH) is produced naturally by fermentation, e.g. in beer- and wine-making,…
A: Given that,volume of flask = 5.0 LPressure of ethylene gas,PC2H4= 1.0 atmPressure of water vapour,…
Q: 49) A particular coal sample contains 3.28 % S by mass. When the coal is burned, the sulfur is…
A: Amount of S in the coal sample=3.28 % S by massAmount of coal burned= 1.2 x 106 lbTemperature = 23…
Q: Choose the resonance structure that results from the arrow pushing scheme below: A H OH Give…
A: The structure given is
Q: You have 50.0mL of 0.520M NH3. Kb = 1.8x10-5 You add 15.0mL of a 0.05M HCl solution. What is the pH…
A: The objective of this question is to calculate the pH of a solution after the addition of a certain…
Q: You react the following molecule with a strong bulky base (like KOtBu), and you know that the Cl…
A: Elimination reactions of alkyl halides occur majorly via two mechanisms. In the E1 mechanism,…
Q: (E) State whether the OVERALL reaction is Oxidation/Reduction or neither for reaction I and II. | 1.…
A: In organic chemistry oxidation and reduction refer to the change in oxidation states. If the…
Q: (i) COOH (h) (1) 2 CH3CH₂Li (2) H3O+ H H3O+ ↑ C=PPh3 + 요. CH3 NILL H+, heat
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Use WILEY Reference to explore the following topics: (CHOOSE ONE that INTERESTS YOU A. DEFINE B.GIVE…
A: Since you have posted question with multiple sub-parts, as per our Q and A guidelines I will solve…
Q: Draw the structure of 3,4-diiodohexane that will be superimposable with its mirror image
A: Molecules can have different spatial arrangements of atoms even if they have the same chemical…
Q: Help pls #9
A: Below complete reaction is Given.Explanation:
Q: Briefly explain the principle and application of potentiometry or pH meter or lon selective…
A: The method of measuring the voltage in a solution between two electrodes is called potentiometry.…
Q: What is the pH of a saturated solution of Zn(OH)2? (Ksp for Zn(OH )2 == 3.0 \times 10-16).
A: Given that,Ksp for pH of the saturated solution=?
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into…
A: The objective of the question is to determine the nature of the reaction (exothermic or…
Q: Assign the absolute configuration of the two chiral centers as R or S. O= OH OH CO₂H
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. We have been…
Q: Calculate the mass, in grams, of 732 atoms of iron, Fe (1 mol of Fe has a mass of 55.85 g).
A: Number of mol of Fe = Number of atoms/Avogadro's number =…
Q: A sample of HI was placed in a container and the following reaction was allowed to come to…
A: Equilibrium constant is defined as the ratio of concentration of products to the concentration of…
Q: (a) Classify the transformation below as oxidation, reduction or neither. (b) Please identify the…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Acetic acid reacts with NaOH. At equilibrium the [CH3COOH] = 0.412 and the [CH3COO-] = 0.247 Ka =…
A: The objective of this question is to calculate the pH of a solution of acetic acid (CH3COOH) and…
Q: Calculations (g) Mass of cool water in cups, mcw (h) Mass of added hot water, mчw 67.5377 g 683542 8…
A: Heat gained or lost (ΔH) by a substance during a temperature change can be calculated using the…
Q: Determine the number of atoms of O in 25.3 moles of Fe(ClO₂)₃.
A: 1 mole of any substance contains 6.022 × 1023 atoms.1 mol = 6.022 × 1023 atoms
Q: 3,3-dichloropropanoic acid Draw the molecule on the canvas by choosing buttons from the Tools (for…
A: The combination of molecular, Lewis, condensed, or skeletal formulas representing the organic…
Q: Suppose a 500. mL flask is filled with 1.7 mol of H₂O, 0.10 mol of CO2 and 0.70 mol of H₂. This…
A: Information about the question
Q: 7. Which bond is correctly labeled with a dipole arrow? + + + C-H O-H N-H C-Cl C-C a. b. C. d. e. a.…
A: The dipole arrow should be from the less electronegative atom to the more electronegative atom.
Q: Will a precipitate (ppt) form when 50.0 mL of 1.1 x 10-3 M Ba(NO3)2 are added to 50.0 mL of 8.4 ×…
A:
Q: You have an aliquot of 11 mL of 0.1093 mol/L phosphoric acid. The ka values for phosphoric acid are…
A: Volume of phosphoric acid = 11 mLMolarity of phosphoric acid sample = 0.1093 mol/LVolume of NaOH…
Q: what's the number-average molecular weight for a linear polytetrafluoroethylene of total length…
A:
Step by step
Solved in 3 steps with 1 images

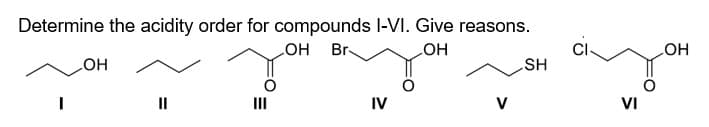
- give clear handwritten solution of all the subparts please!Rank in order of Increasing Bronsted acidity the structures I, II and III.Suppose you made a mistake in Ag group analysis, If instead of 6M HCl you used 6M H2SO4 what will you observe? Can you continue from that point on to separate the cations ?How?Explain
- Provide solution and explanation for the problem below.The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Questions: (i) Give the name and suggest the colour of the precipitate B. Hence explain why it is necessary to recrystallize B several times. (ii) Why was it necessary to obtain a constant melting point for B?The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. 1g of phenylamine yielded 1.2g of compound A. Calculate the percentage yield of the reaction.
- The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Why was it necessary to obtain a constant melting point for B?The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Give the structure of the product formed when an acidified solution of compound A is reacted with (i)Naphthalen-2-ol (2-naphthol) (ii) Sodium cyanideThe following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Question: Answer both (i) and (ii) below (i) Give the structure of the product formed when an acidified solution of compound A is reacted with Naphthalen-2-ol (2-naphthol) and Sodium cyanide separately (ii) 1g of phenylamine yielded 1.2g of compound A. Calculate the percentage yield of the reaction.
- The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Write the equation for the reaction between compound A and phenol Give the name and suggest the colour of the precipitate B. Why is it necessary to recrystallize B several times?The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. (a) What is the general name given to the reaction between phenylamine and sodium nitrite and explain why it's necessary to carry out the reaction at low temperatures. (b) Write the equation for the reaction between compound A and phenolThe following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. What is the general name given to the reaction between phenylamine and sodium nitrite and explain why it's necessary to carry out the reaction at low temperatures? Write the equation for the reaction between compound A and phenol Write the equation for the reaction between phenylamine and sodium nitrite.



