Determine the pH of a buffer that is 0.95 M HBRO and 0.68 M KBRO. The value of pKa for HBRO is 8.68. 3 NEXT Based on the given values, set up ICE table in order to determine the unknown. H,Oʻ(aq) BrO(aq) + HBRO(aq) H,O() Initial (M) Change (M) Equilibrium (M) 5 RESET +2x 0.95 0.68 8.68 +x 0.95 - x 0.95 - 2x 0.68 + x 0.68 - x 0.68 + 2x -2x 0.95 + x 0.95 + 2x 0.68 - 2x
Determine the pH of a buffer that is 0.95 M HBRO and 0.68 M KBRO. The value of pKa for HBRO is 8.68. 3 NEXT Based on the given values, set up ICE table in order to determine the unknown. H,Oʻ(aq) BrO(aq) + HBRO(aq) H,O() Initial (M) Change (M) Equilibrium (M) 5 RESET +2x 0.95 0.68 8.68 +x 0.95 - x 0.95 - 2x 0.68 + x 0.68 - x 0.68 + 2x -2x 0.95 + x 0.95 + 2x 0.68 - 2x
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter16: Solubility And Precipitation Equilibria
Section: Chapter Questions
Problem 9P
Related questions
Question
I really need help with this review problem
thanks
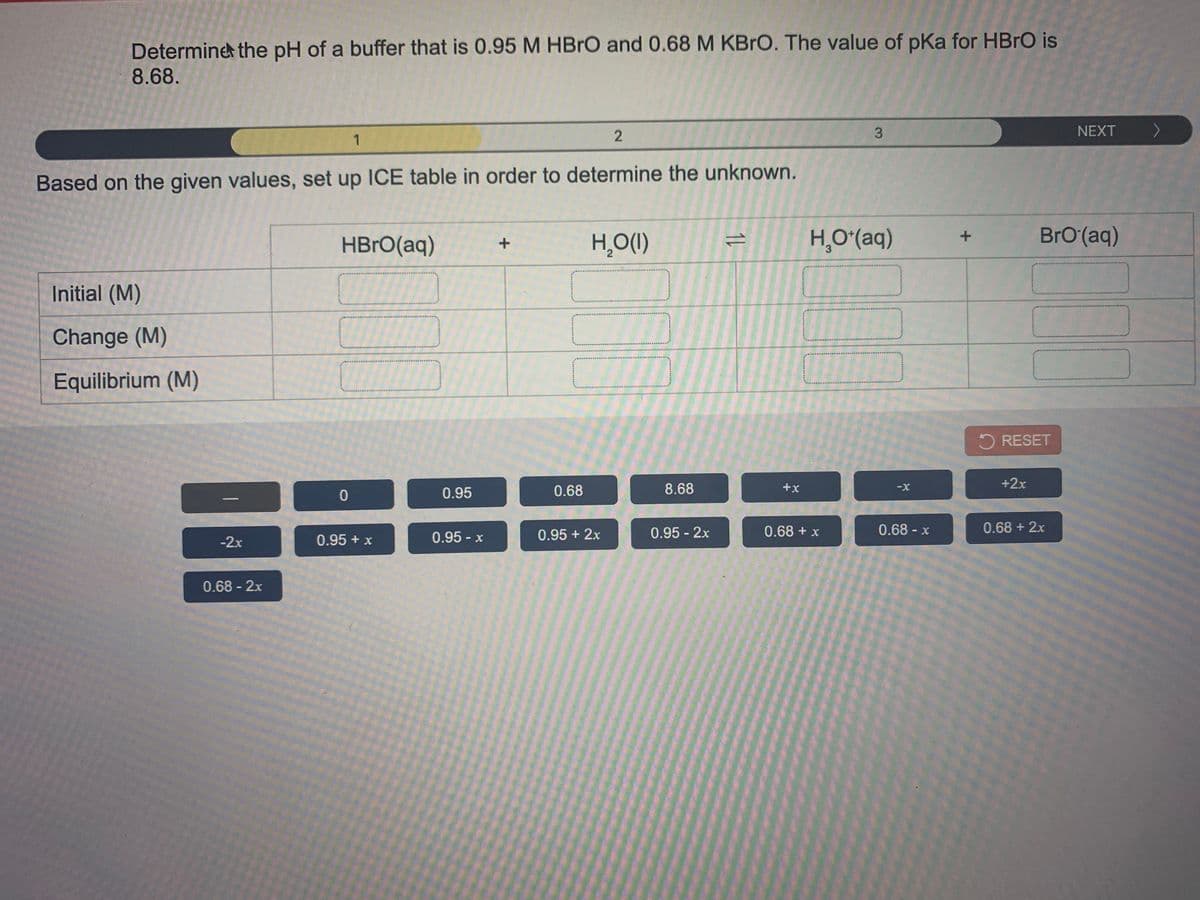
Transcribed Image Text:Determine the pH of a buffer that is 0.95 M HBRO and 0.68 M KBRO. The value of pKa for HBRO is
8.68.
3.
NEXT )
>
Based on the given values, set up ICE table in order to determine the unknown.
HBrO(aq)
H̟O(1)
H̟Oʻ(aq)
BrO(aq)
Initial (M)
Change (M)
Equilibrium (M)
5 RESET
0.68
8.68
+x
+2x
0.95
0.95 + 2x
0.95 2x
0.68 + x
0.68- x
0.68 + 2x
-2x
0.95 + x
0.95- x
0.68 - 2x
![Determine the pH of a buffer that is 0.95 M HBrO and 0.68 M KBRO. The value of pKa for HBRO is
8.68.
3.
NEXT
K PREV
Based on your ICE table and definition of Ka, set up the expression for Ka in order to determine the unknown. Do not
combine or simplify terms.
Ka
O RESET
[0]
[0.95]
[0.68]
[8.68]
[x]
[2x]
[2x]?
[0.95 + x]
[0.95 - x]
[0.95 + 2x]
[0.95 - 2x]
[0.68 + x]
[0.68 - x]
[0.68 + 2x]
[0.68 - 2x]
8.68
0.939
4.79 x 106
2.09 x 109](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F5c6d7610-fae0-4fdd-8afb-1e0bd9bb140b%2Fecc5a6c2-b3a9-4861-9362-0889b3be5bb1%2Fuc6ryht_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Determine the pH of a buffer that is 0.95 M HBrO and 0.68 M KBRO. The value of pKa for HBRO is
8.68.
3.
NEXT
K PREV
Based on your ICE table and definition of Ka, set up the expression for Ka in order to determine the unknown. Do not
combine or simplify terms.
Ka
O RESET
[0]
[0.95]
[0.68]
[8.68]
[x]
[2x]
[2x]?
[0.95 + x]
[0.95 - x]
[0.95 + 2x]
[0.95 - 2x]
[0.68 + x]
[0.68 - x]
[0.68 + 2x]
[0.68 - 2x]
8.68
0.939
4.79 x 106
2.09 x 109
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
