Q: NiCl2 + 6 NH3 + 2 KClO4 ® Ni(NH3)6(ClO4)2 + 2 KCl if you mix 1.0406 g of NiCl2, 6.0092 g NH3 and 2.0...
A: Given, NiCl2 + 6 NH3 + 2 KClO4 Ni(NH3)6(ClO4)2 ...
Q: Calculate the activity of IO3- in a saturated solution of La(IO3)3 with 0.15 M Al(NO3)3.
A: Given, the activity of IO3- in a saturated solution of La(IO3)3 with 0.15 M Al(NO3)3.
Q: Caffeine, a stimulant found in coffee, contains 49.5% C, 5.15% H, 28.9% N, and 16.5% O by mass and h...
A: First we calculate the emperical formula . Then claculate n with the help of molar mass as given in ...
Q: Identify the peaks and structure.
A: NMR is the nuclear magnetic resonance spectroscopy that falls under the radio frequency region of el...
Q: Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2Al(s) + 3C1...
A:
Q: Problem 48 - IR spectrum Problem 48 C10H200 40- MW = 156 35- 30- 25- 20- 15- CDC1; 10- 3500 3000 250...
A:
Q: PLEASE HELP DRAW AND WRITE THE REACTION, EQUATIONS AND MECHANISMS FOR THE REACTION OF CYCLOHEXENE WI...
A: Alkene reacts with Br2 in presence of water to give halohydrin.
Q: 4) Batholiths have: A- regular forms B- Irregular forms C-Sometime regular forms
A: As per the rules only the first question can be answered.
Q: 1. How many MOLES of boron trichloride are present in 4.21 grams of this compound ? moles. 2. How ma...
A:
Q: Determine the mass %, molality and molarity of a solution formed by dissolving 12g of calcium hydrox...
A:
Q: At 500 ∘C∘C, cyclopropane (C3H6) rearranges to propene (CH3−CH=CH2). The reaction is first order, an...
A: First-order reaction: The rate of the first-order reaction depends only on one reactant. According t...
Q: 114
A: The first step is to count the number of each species present in HX, HY and HZ solutions and presen...
Q: At a certain temperature the rate of this reaction is first order in N,O, with a rate constant of 0....
A: The given reaction follows the first-order kinetics. The rate of the first-order reaction depends on...
Q: What is the oxidation state for KMnO4? Please show or explain calculation
A:
Q: Br2 (1eq) H3CO- CH3 FeBr3
A:
Q: Q1.Fill in positive, negative and neutral The isoelectric point, pl, of two amino acids is shown bel...
A:
Q: Object A has a charge of 3.6 × 10 °C and object B has a charge of 4.7 × 10 °C. What is the electric ...
A:
Q: A. What is the molarity of the barium hydroxide solution? M This barium hydroxide solution is then u...
A: Given mass of potasium hydrogen phthalate =0.945g Volume =34.3mL
Q: Solid aluminum and oxygen gas combine in a synthesis reaction. 4.80mol of powdered aluminum is place...
A: Given, Moles of aluminium (Al) = 4.80 mol Moles of Oxygen gas (O2) = 3.00 mol Mass (in grams) of Alu...
Q: "Each pi-l c-ntains 12.5 - of act-ve -ngre-dient. Adm-i--ister thr-e tim-s -aily unt-l ymptoms dimi-...
A: According to Charle's law, volume of a gas is directly proportional to temperature of that gas if pr...
Q: Friedel-Crafts
A:
Q: reaction quotient
A:
Q: ne number of grams of krypton in th
A: In this question we have to determine the number of grams of Krypton in the mixture with above infor...
Q: The pH of pure water at 50°C. is 6.63. At 75°C the pH is 6.35. Compare Kw values at 25°C, 50.°C and...
A: Answer attached below
Q: Liquids and gases take the following characteristic(s) of their containers. Which is/are it? volume...
A: Both Gas and liquid molecules have large intermolecular separation. Among liquid and gas molecules,...
Q: Calculate the concentration of iodide ions (1) in a saturated solution of lead iodide, Pbl2 (Ksp = 7...
A:
Q: Problem 19 - IR spectrum Problem 19 C,H,NO4 MW = 193 25- MSO-de 15- MSO-de 10- 3500 3000 2500 2000 1...
A: IR spectra provides the information about the functional groups of unknown compound.
Q: Item 1 Part E Indicate whether each of the compounds is a primary, secondary, or tertiary alcohol. D...
A:
Q: A 0.50 mL human saliva sample was placed in a 100-mL volumetric flask and was diluted to mark with b...
A: Given that, a 0.50 mL of human saliva sample was placed in a 100 mL volumetric flask and was diluted...
Q: What is the final glycosidic link to be broken during glycogen debranching? a-1,3-glycosidic linkage...
A:
Q: When liquid phosphorus trichloride is added to water, it reacts to form aqueous phosphorous acid, H3...
A: • Liquid phosphorus trichloride on reaction with water gives aqueous phosphorus acid and aqueous hyd...
Q: Choose a balanced chemical equation for the reaction of molecular oxygen with beryllium metal to for...
A:
Q: Part B How many grams of NH3 can be produced from 3.73 mol of N2 and excess H2 . Express your answer...
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product ...
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Consider the following exothermic reaction: C2H4 (9) + Cl2 (9) = C2H¾C12 (g) Part A If you were a ch...
A:
Q: To treat a burn on his hand, a person decides to place an ice cube on the burned skin. The mass of t...
A: Please find your solution below : Thermochemistry is a branch of chemistry in which we measure the a...
Q: The solubility of Na2SO4 will probably increase with increasing temperature. A) True B) False
A: False
Q: GUse curved arrows to show the mechanism of the step below. Make the ends of your arrows specity the...
A: Mechanism of the step Product of the reaction
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate cons...
A: A zero order reaction is the independent of the reactant concentration. And formula used [A]t = - k...
Q: Consider the reaction 2H2(g) + O2 (g) = 2H2O(g) How will the equilibrium shift when hydrogen gas is ...
A: HSAB concept- Hard- Hard And Soft-Soft interactions are strong.
Q: New Ta Grades | Schoology 9 Schoology 9 Practice: Vertex & Factored Form x b learn.rdale.org/common-...
A:
Q: How many electrons in an atom could have these sets of quantum numbers? n = 3 18 electrons n = 5, € ...
A:
Q: How many molecules of water occur per formula unit of MgSO4. Write the complete formula of the hydra...
A:
Q: Atomic number
A: The atomic number is generally use to uniquely identifies a chemical element.
Q: Write a hypothesis with a in detail reason for the testable question? Testable Question : Does the a...
A: We have to determine that Does the amount of baking soda effect the circumference of the balloon.
Q: 50.050.0 mL solution of 0.1390.139 M KOHKOH is titrated with 0.2780.278 M HClHCl. Calculate the pH o...
A:
Q: Predict the product of the oxidation of each compound below. If the compound is oxidized one time, w...
A:
Q: ach alcohol below to its proper category (primary, secondary or tertia CH3 OH
A:
Q: The rate of a certain reaction is given by the following rate law: rate =k[N,][H,] Use this informat...
A: Given :- rate = K[N2] [H2]2 To determine :- Order with respect to N2,H2 and also determine the ov...
Q: 1. Give a full description (type -purine/pyrimidine- name adenine/guanine/uracil/thymine/cytosine, w...
A: This question is related to biomolecules. Bases are divided in to two categories Purines Pyrimidine
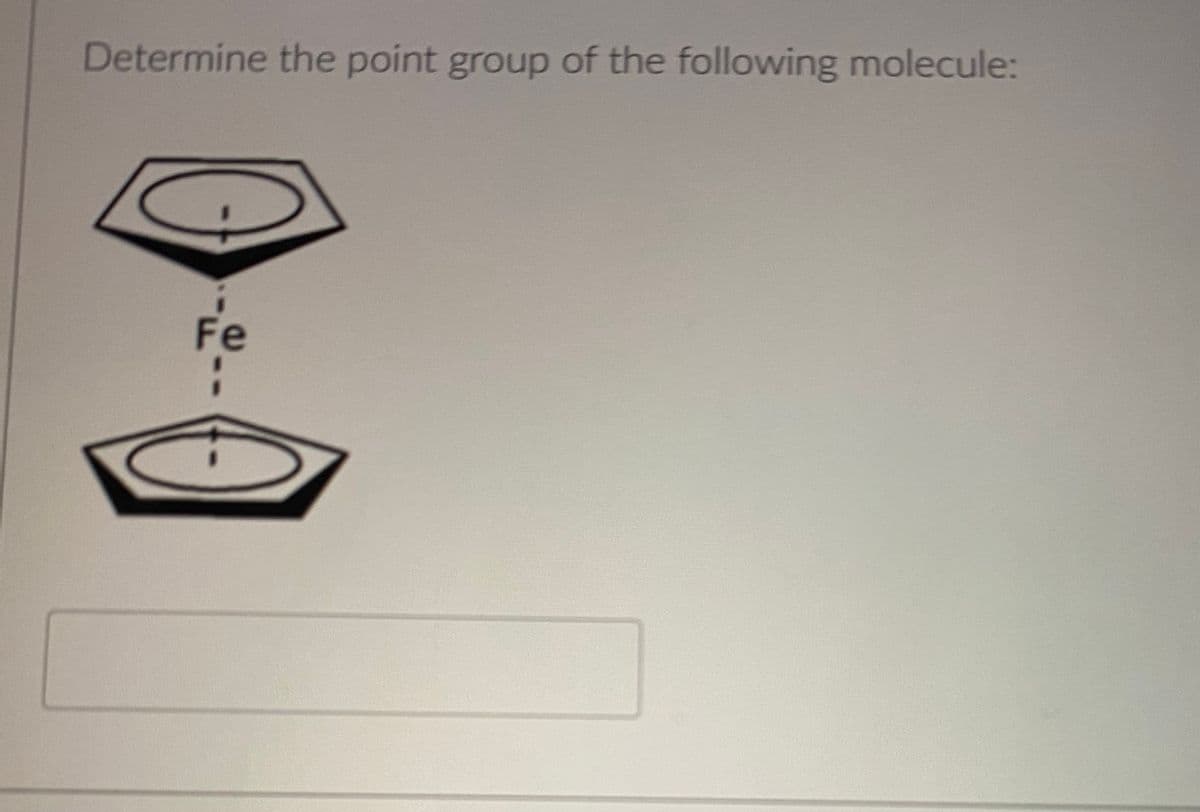
Step by step
Solved in 2 steps

- Determine the point groups of the following molecules. a Fe(CO)5, which has a trigonal bipyramidal structure, b CO32, which has three resonance structures that contribute to its overall shape, c The perfectly staggered conformation of ethane, d The perfectly eclipsed conformer of ethane.Structural isomers can have very different point groups. There are three isomers of dichlorobenzene. Identify the point groups of the three isomers.Structural isomers can have very different point groups. Determine the point groups of 1,4cyclohexadiene and 1,3cyclohexadiene, which both have the molecular formula C6H8.
- Determine the point groups of the following molecules. a Hydrogen selenide, H2Se b Partially deuterated hydrogen sulfide, or HDS c The chair conformer of cyclohexane, C6H12 d The boat conformer of cyclohexane, C6H12Construct the symmetry-adapted linear combination molecular orbitals for hydrogen sulfide, H2S.What are the number of classes and the order of the following point groups? a C2v b D2h c D6h d S4 e Cs.
- Determine which single symmetry operation of the following point groups is equivalent to the given combination of multiple symmetry operations. a In C2v, C2v=? b In C2h, iC2=? c In D6h, C6h=? d In D2d, C2C2=? e In Oh, iS4=?Figure 13.27 shows the structure of the molecule porphine. Figure 13.27 The structure of porphine. Determine the symmetry elements present in the molecule, and its point group. Does the point group change if an Fe2+ ion is substituted for the two hydrogen atoms in the center of the porphine ring?Show that any two of the irreducible representations of the following point groups are orthogonal to each other. a C2 b C2v c D2h d Oh e Td
- Determine the symmetry species of the D3h point group for the sp2 hybrid orbitals, assuming that the C3 axis is coincident with the z-axis and that one of the orbitals lies along the positive x-axis. See Example 13.16.Identify the symmetry elements present in the following objects. a A ream of blank paper, no holes. b A ream of blank three-holed paper. c A round pencil, unsharpened, with cylindrical eraser. d A round pencil, sharpened, with cylindrical eraser.In your own words, explain why an object that has more symmetry elements is said to have higher symmetry than an object with fewer symmetry elements.

