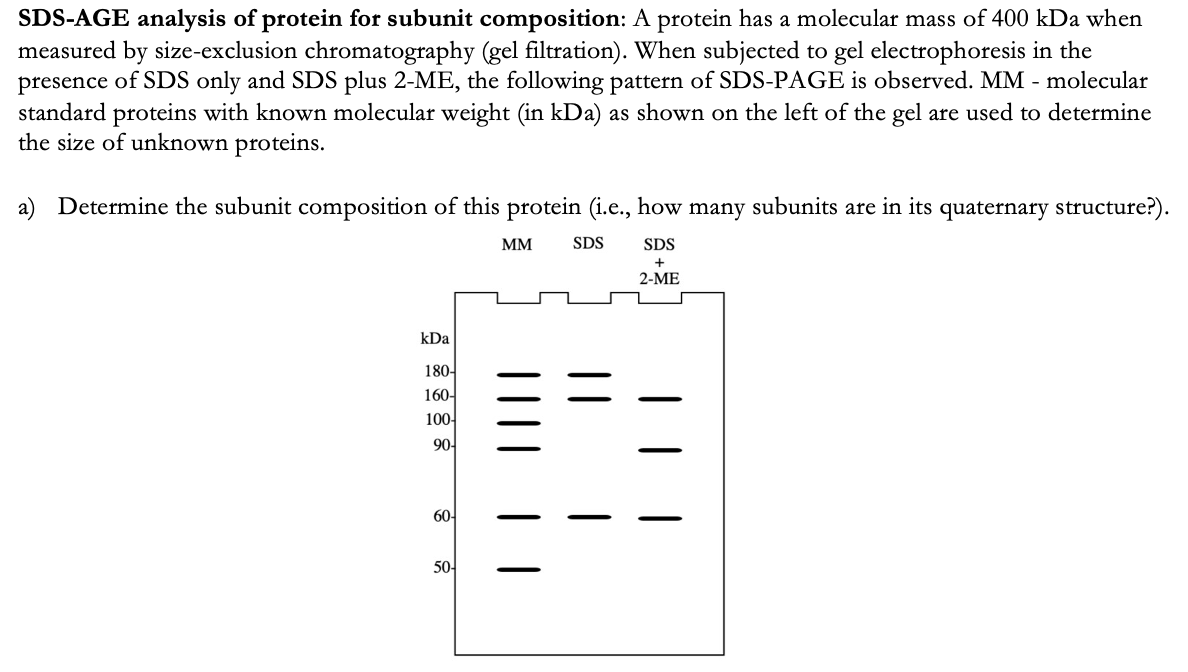
Determine the subunit composition of this protein (i.e., how many subunits are in its quaternary structure?). MM SDS SDS
Q: A peptide with the sequence isoleucine1-aspartate2-valine3-lysine4-proline5-glutamate6 is located at…
A: Amino acids combine to form proteins by the formation of peptide bond. Amino acids have a one amino…
Q: Explain briefly why the ionizable properties of amino acids are so crucial in the structure and…
A: Introduction Amino acids are a kind of nutrient. Proteins are polymers of nitrogenous chemicals…
Q: List two types of DNA mutations and state which one is more likely to have a severe effect on the…
A: DNA mutations are a sudden change in the DNA sequence. Mutations can be caused by various factors,…
Q: Two peptide segments are shown below. Predict which one would have the most negative AG when going…
A: Protein folding is a spontaneous reaction by which most of the proteins fold inside the cell without…
Q: Draw the structure of the following polypeptide at physiological pH. You do not need to indicate the…
A: Protein is composed of various amino acids in various proportions, which determine the structure…
Q: Explain the importance of primary and tertiary structures in the functioning of a protein.
A: Proteins are one of the important bio-molecules which are composed of different amino acids. The…
Q: , calculate the average amino acid residue weight in a protein of typical composition. This is a…
A: Proteins are composed of the linear chain of amino acids attached via peptide bonds. Proteins are…
Q: Hydrogen bonds and hydrophobic interactions play important roles in stabilizing and organizing…
A: Protein is an important biomolecule made up of small units called amino acids. These proteins are…
Q: Describe the synthesis of a protein from amino acids. Then describe the primary, secondary,…
A: Proteins are the building blocks of body. Proteins are required for the growth and repair of body.
Q: All the necessary information to form the three-dimensional structure of the protein is provided by:…
A: Proteins are formed by amino acid residues. These amino acid residues are held together by peptide…
Q: Proteins have a variety of functions within a living cell.what are the possible functions of…
A: Proteins contain amino acids and these amino acids form long peptide chains.
Q: A polypeptide has a high pI value. Suggest which amino acid residues might comprise it.
A: The isoelectric point (pI) is the pH of a solution at which the net charge of a protein becomes…
Q: How many N-terminus are there in this cartoon?
A: *There are four types of protein structure are present. primary structure secondary structure…
Q: Protein folding
A: Proteins are composed of some 20 types of amino acid residues, and they would be more or less alike…
Q: The detergent sodium dodecyl sulfate (SDS) denatures proteins. Suggest how SDS destroys protein…
A: The protein or peptide chain is folded into a complex structure with the help of several kinds of…
Q: Describe in no more than ten sentences the four levels of protein structure, and cite the…
A: Proteins are composed polypeptide chain, which is bonded with the peptide bond.
Q: Which of the following statements about protein folding and structure are true? Check all that…
A: Molecular chaperones interact with unfolded or partially folded protein subunits, and help them in…
Q: Compare primary, secondary, tertiary and quaternary structures of protein
A: Proteins are composed of carbon, nitrogen, hydrogen, oxygen, and sulfur atoms. Proteins are…
Q: Theoretically, a protein could assume avirtually infinite number of configurations and…
A: Proteins are made up of amino acids. They consists of 4 major structures- primary, secondary,…
Q: Describe in no more than ten sentences the four levels of protein structure, and cite the…
A: Proteins are defined as complex, compounds that are organic in nature. They are composed of many…
Q: Describe in detail the vapour diffusion method for crystallising a protein
A: Vapor diffusion is the most typically employed method of protein crystallization. In this method,…
Q: Describe how a polypeptide can fold to become a functioning protein. Be sure to address the four…
A: A peptide bond connects each amino acid to its neighbors that are translated from the sequence of…
Q: Of the following amino acids, which 3 are most likely to be found on surface of a single protein.…
A: Proteins are the ultimate products of the genes. DNA is transcribed into m RNA and this is…
Q: The function of proteins can be regulated in a number of ways. What is cooperativity, and how does…
A: Proteins are massive biomolecules and macromolecules made up of one or more long chains of amino…
Q: Protein structure.Circle one of the three amino acid sequences that is most likely to form a stable…
A: The common secondary structure of the protein are alpha-helix and beta-sheets. The alpha-helix are…
Q: An intrinsically disordered protein (IDP) is a protein that lacks a fixed three- dimensional…
A: Given; Freely joined chain length of a single amino acid = 3Å Total number of constituent amino…
Q: The use of salt bridges or hydrophobic interactions (or pockets) to stabilize interactions between…
A: Proteins: Proteins are biological polymers and mainly made up of amino acids those that are linked…
Q: Discuss the different structures of protein. What are the five factors that promote protein folding…
A: Introduction : Proteins are large, biologically active molecules that play a number of important…
Q: The use of salt bridges or hydrophobic interactions (or pockets) to stabilize interactions between…
A: The quaternary structure of a protein is the association of several protein chains or subunits into…
Q: D arrangement of localized regions of proteins A. PRIMARY B. SECONDARY
A: The three-dimensional arrangements of atoms in an amino acid chain, that folded up into specific…
Q: The three-dimensional structure of a protein is determined by its primary, secondary, and tertiary…
A: Protein is the biomolecule which comprise the one or more long chain of amino acids which belongs to…
Q: Discuss the different structures (primary, secondary, tertiary, and Quaternary structures) of…
A: Proteins are amino acid polymers. The sequence of amino acids determines their three-dimensional…
Q: Proteins can be separated into 9 general classifications based on the role they play in a cell. List…
A: Proteins can be classified into following types:- Fibrous Proteins Globular Proteins Derived…
Q: A protein that is normally found in an aqueous solution has these amino acids in its primary…
A: Proteins are composed of different amino acids with different chemical nature. Amino acids are…
Q: If you incubate a protein with a single beta sheet in solution at pH = 14, the beta sheet no longer…
A: Proteins are essential for the normal functioning of the body. They are involved in providing…
Q: To examine: Whether the statement "Loops of polypeptide that protrude from the surface of a protein…
A: Proteins are essential dietary components. It is made up of a polypeptide chain, which is a chain of…
Q: a peptide has the sequence: Asp-His-Tyr-Cys. What is the net charge on this molecule at pH 7.0? Will…
A: Given, Asp-His-Tyr-Cys thus considering Asp at the amino terminal and Cys at the carboxylic terminal…
Q: How can a protein’s potential function be determined from a protein’s primary structure?
A: In a protein, the primary structure refers to the sequence of the amino acids in the polypeptide…
Q: Protein Structure Describe the four levels of protein structure: Primary, secondary, tertiary, and…
A: Proteins are the most plentiful natural molecules of the living framework. They happen in all…
Q: Which intermolecular forces are important in acetic acid, CH3 –(C=0)-oh?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Levels of structure in a protein (primary, secondary, tertiary, quaternary): What each of these…
A: Protein is a biomolecule consisting of amino acids joined together via peptide bond. Each amino acid…
Q: V-B. Which of the following peptides would be more soluble at the indicated pH? 1. (Gly)20 or…
A: There are two amino acids that are classified into 4 classes: 1. Nonpolar(aliphatic and aromatic) 2.…
Q: Protein denaturation disrupts the noncovalent interactions in a protein. Which level of protein…
A: By subjecting proteins or nucleic acids to an external stress or substance such as a strong acid or…
Q: Explain the difference between the primary, secondary, tertiary, and quaternary structures of a…
A: Protein: They are large bio or macro molecules which contains one or more long chains of amino acid…
Q: Many plasma proteins found in an aqueous environment are globular in shape. Which amino acid side…
A: The outer surfAce of the globular protein is coated with hydrophilic protein is coated side chains,…
Q: Draw the tripeptide FTQ, making sure to care for stereochemistry. * Identify the N-terminus and the…
A: Hi! Thanks for your question. As 'FTQ' is an unspecified term we are answering the question with…
Q: At pH = 7, the small peptides DE, LR and VF are separated using anion exchange chromotography, which…
A: Anion exchange chromatography is a form of ion-exchange chromatography which is used to separate…
Q: Can you please describe the specific components that each of these structures are comprised of (…
A: Proteins are the biomolecules which are made up of amino acids. Amino acids are joined together with…
Q: Decide if the side chains of the following peptides are nonpolar or polar,and label the hydrophobic…
A: Amino acids are polymers of proteins that consist of the carboxyl group, amino group, and an R group…
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

- Denaturation of Proteins The following are four levels of protein structures. Identify the inter- and intramolecular forces of attractions that stabilized each level. Level of Protein Structure Forces of Attraction Present Primary Secondary Tertiary Quaternary 2.) What is denaturation? 3.) What are denaturing agents? Give three examples and describe their effects on protein. 4.) Differentiate reversible and irreversible denaturation. 5.)Why is 70% ethanol is more effective than 100% ethanol to kill bacteria?Purification of a new unknown protein that you isolated from tissue and Assume that you have reached the following data during the characterization; Gel filtration: Gel filtration in protein native conformation When chromatographed, it has a molecular weight of 240000 daltons (240 kDa) is detected to be around. Gel filtration: The same protein is first denatured with 6 M guanidinium hydrochloride subjected to gel filtration chromatography again under denatured conditions. is retained, and the only column from the column with a molecular weight of about 60000 daltons (60 kDa) a protein is obtained. SDS-PAGE: Protein finally SDS-PAGE in the presence of beta-mercaptoethanol (Sodium dodecyl-sulphate polyacrylamide gel electrophoresis) analysis being held. As a result of SDS-PAGE analysis, their weight in the gel is approximately 40000 daltons. Two protein bands corresponding to (40 kDa) and 20000 daltons (20 kDa) is observed. In the light of these findings, the quaternary/quaternary…Shape and dimension. (a) Tropomyosin, a 70-kDa muscle protein, is a two-stranded α-helical coiled coil. Estimate the length of the molecule. (b) Suppose that a 40-residue segment of a protein folds into a two-stranded antiparallel β structure with a 4-residue hairpin turn. What is the longest dimension of this motif?
- Avidin exists as a protein complex of around 68 kDa. Research to determine the types of interactions that hold the avidin complex together. Based on your research, which molecular weight would you expect for avidin when treated with beta-mercaptoethanol and run through a native gel? 35 kDa 68 kDa 10 kDa 19 kDaUnderstanding the Relevance of Chaperones in Protein Folding Protein molecules, like all molecules, can be characterized in terms of general properties such as size, shape, charge, solubility/hydrophobicity. Consider the influence of each of these general features on the likelihood of whether folding of a particular protein will require chaperone assistance or not. Be specific regarding just Hsp7O chaperones or Hsp7O chaperones and Hsp60 chaperonins.The Energetic Cost of Peptide Elongation How many ATP equivalents are consumed for each amino acid added to an elongating polypeptide chain during the process of protein synthesis?
- Predict the number of bands and apparent mol. wt. of the following proteins on SDS gels. 1. A trimeric protein containing three chains, each with a molecular weight of 60,000 Da (60 kDa).1CFD CALCIUM-FREE CALMODULIN How long is the protein’s primary sequence?Does this protein have any secondary, tertiary and/or quaternary structures present?Are there any molecular additions to this protein that are not amino acAla-Arg-Val-His-Asp-Gln Given the polypeptide chain above Estimate the net charge of the polypeptide chain at physiological pH (7.4) and at pH 5.0 . How many peptide bonds are there? What kind of polypeptide is it?
- Vertebrate proteins? What is meant by the term polypeptide backbone?Some proteins migrate anomalously in SDS-PAGE gels. For instance, the molecular weight determined from an SDS-PAGE gel is sometimes very different from the molecular weight determined from the amino acid sequence. Suggest an explanation for this discrepancy.Peptide mass determination. You have isolated a proteinfrom the bacterium E. coli and seek to confirm its identityby trypsin digestion and mass spectrometry. Determinationof the masses of several peptide fragments has enabled youto deduce the identity of the protein. However, there is adiscrepancy with one of the peptide fragments, whichyou believe should have the sequence MLNSFK and an(M 1 H)1 value of 739.38. In your experiments, yourepeat edly obtain an (M 1 H)1 value of 767.38. What isthe cause of this discrepancy and what does it tell youabout the region of the protein from which this peptide isderived?

