Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the solubility constant expression, and solving the expression. Complete Parts 1-2 before submitting your answer. 1 2 NEXT The molar solubility of Mg(CN), is 1.4 x 105 M at a certain temperature. Fill in the ICE table with che appropriate value for each involved species to determine concentrations of all reactants and products at this temperature... Mg(CN),(s) Mg²"(aq) 2CN'(aq) + Initial (M) Change (M) Equilibrium (M) 5 RESET 1.4 x 10* -1.4 x 10* 2.8 x 10* -2.8 x 10 +2x +x -2x 1.4 x 10* + x 1.4 x 10* + 2x 1.4 x 10* - x 1.4 x 10* - 2x 2.8 x 10 + x 2.8 x 10 + 2x 2.8 x 10* -x 2.8 x 10* - 2x MAR 31 stv J MacBook Pro 1L
Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the solubility constant expression, and solving the expression. Complete Parts 1-2 before submitting your answer. 1 2 NEXT The molar solubility of Mg(CN), is 1.4 x 105 M at a certain temperature. Fill in the ICE table with che appropriate value for each involved species to determine concentrations of all reactants and products at this temperature... Mg(CN),(s) Mg²"(aq) 2CN'(aq) + Initial (M) Change (M) Equilibrium (M) 5 RESET 1.4 x 10* -1.4 x 10* 2.8 x 10* -2.8 x 10 +2x +x -2x 1.4 x 10* + x 1.4 x 10* + 2x 1.4 x 10* - x 1.4 x 10* - 2x 2.8 x 10 + x 2.8 x 10 + 2x 2.8 x 10* -x 2.8 x 10* - 2x MAR 31 stv J MacBook Pro 1L
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.60PAE
Related questions
Question
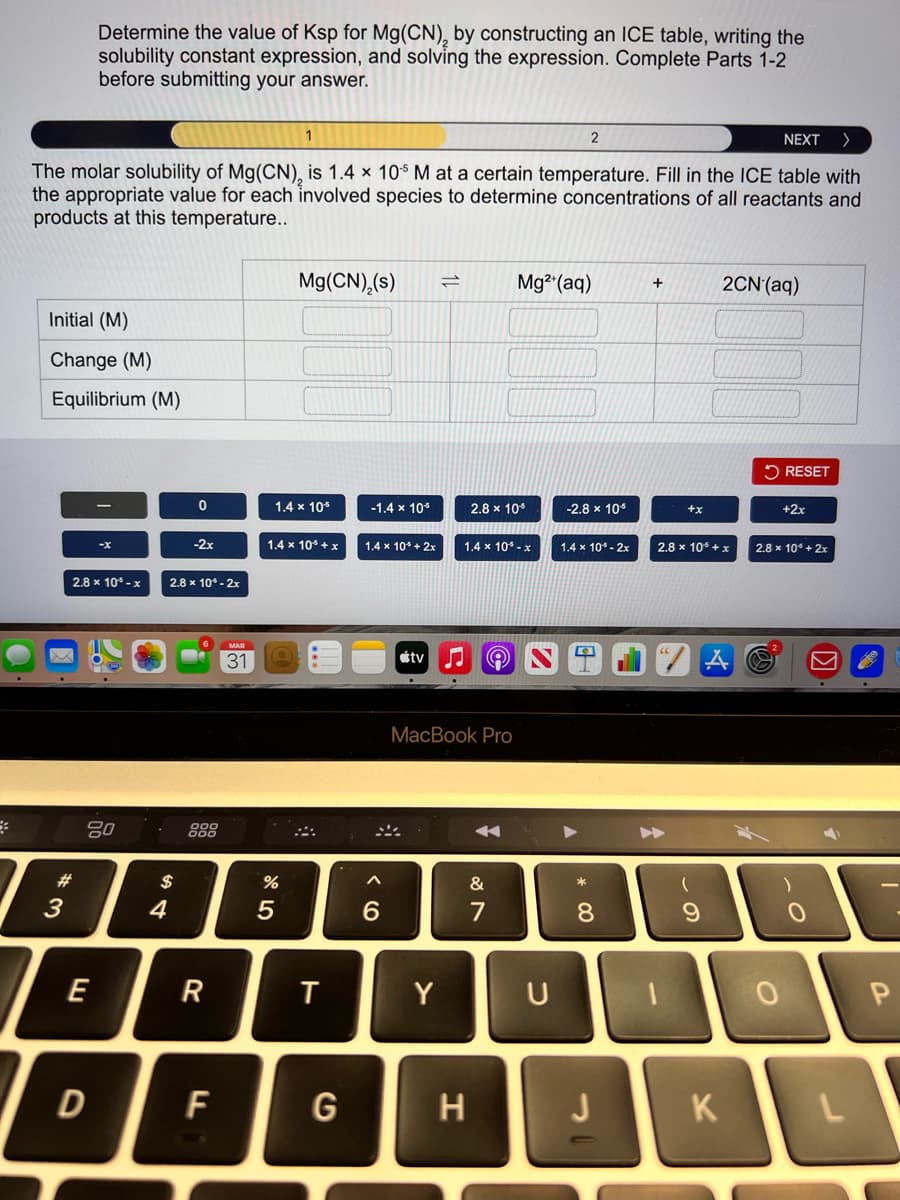
Transcribed Image Text:Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the
solubility constant expression, and solving the expression. Complete Parts 1-2
before submitting your answer.
NEXT
The molar solubility of Mg(CN), is 1.4 × 10$ M at a certain temperature. Fill in the ICE table with
the appropriate value for each involved species to determine concentrations of all reactants and
products at this temperature..
Mg(CN),(s)
Mg2 (aq)
2CN'(aq)
+
Initial (M)
Change (M)
Equilibrium (M)
2 RESET
1.4 x 10*
-1.4 x 105
2.8 x 104
-2.8 x 10*
+x
+2x
-2x
1.4 x 10* + x
1.4 x 10* - 2x
2.8 x 10 +x
-X
1.4 x 10* + 2x
1.4 x 10* - x
2.8 x 10° + 2x
2.8 x 10* -x
2.8 x 10* - 2x
31
étv
MacBook Pro
80
888
#
$
*
3
4
5
7
8
E
R
Y
U
D
G
H
J
K
* C0
![Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the
solubility constant expression, and solving the expression. Complete Parts 1-2
before submitting your answer.
く
PREV
1
Using the values from the ICE table, construct the expression for the solubility constant. Each
reaction participant must be represented by one tile. Do not combine terms Once the expression
is constructed, solve for Ksp..
Ksp
2 RESET
[0]
[1.4 × 10°]
[2.8 x 10°
(1.4 x 10*j
[2.8 x 10°
[x]
[2x]?
[1.4 x 10 + x]
[1.4 x 10* + 2x
[1.4 x 10* - x]
[1.4 x 10* - 2x]
[2.8 x 10* + x]
[2.8 x 10* + 2x)
[2.8 x 10* - x]
[2.8 x 10* - 2x
1.4 x 10
2.7 x 101
1.1 x 101*
2.2 x 10"
3.9 x 1010
MAR
31
tv
MacBook Pro
888
$
&
3
4
6.
7
8
E
R
T
Y
U
D
F
H
J
K](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fdbf8765a-c0ab-46fe-8663-61e338d72b7c%2Ff8eb0a7b-1ddb-4ca4-8cf1-d5c705b1850e%2Fs6toomt_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the
solubility constant expression, and solving the expression. Complete Parts 1-2
before submitting your answer.
く
PREV
1
Using the values from the ICE table, construct the expression for the solubility constant. Each
reaction participant must be represented by one tile. Do not combine terms Once the expression
is constructed, solve for Ksp..
Ksp
2 RESET
[0]
[1.4 × 10°]
[2.8 x 10°
(1.4 x 10*j
[2.8 x 10°
[x]
[2x]?
[1.4 x 10 + x]
[1.4 x 10* + 2x
[1.4 x 10* - x]
[1.4 x 10* - 2x]
[2.8 x 10* + x]
[2.8 x 10* + 2x)
[2.8 x 10* - x]
[2.8 x 10* - 2x
1.4 x 10
2.7 x 101
1.1 x 101*
2.2 x 10"
3.9 x 1010
MAR
31
tv
MacBook Pro
888
$
&
3
4
6.
7
8
E
R
T
Y
U
D
F
H
J
K
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning