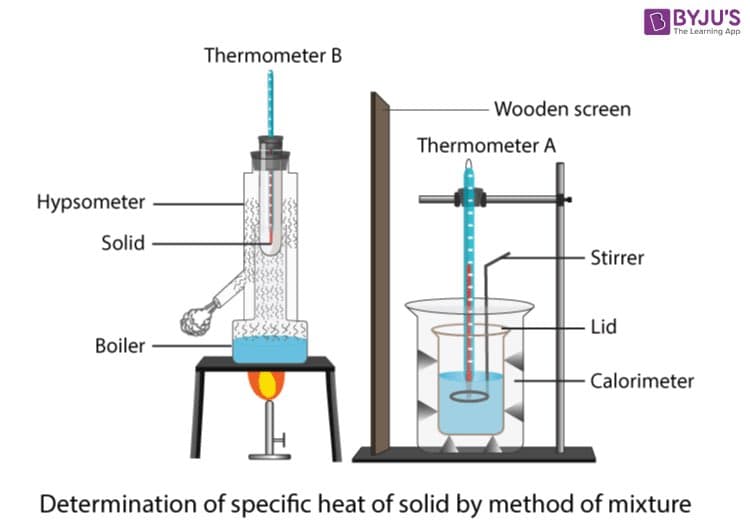
Directions: Draw the experimental set up for the determination of heat of an unknown metal. Any missing answer will result in DISLIKING your solutions.
Q: Thank you, but why was 0.0135 multiplied by 2?
A: Answer: For most of the reaction on increasing the temperature rate of reaction increases because…
Q: Ice at 0.0 °C is placed in a Styrofoam cup containing 381. g of a soft drink at 15.8 °C. The…
A: To determine the mass of ice that has melted, first of all, the energy that has been liberated by…
Q: How would yo perform the experiment of iron sulphide safely in a controlled manner?
A: Performing an experiment involving the formation of iron sulfide (FeS) can be done in a controlled…
Q: Use the following equation to answer the following question: 3Ba(NO3)2+Al2(SO4)3=3BaSO4+2Al(NO3)3…
A: Using balanced chemical equation and stoichiometry, mass of aluminum sulfate can be calculated to…
Q: How many moles are present in 20.29 grams of X? (Assume that the molar mass of X is 32.95 grams per…
A: Mass of X = 20.29 g Molar mass of X = 32.95 g/mol To determine number of moles present of X
Q: B(OR)3 i ?
A: This reaction is nucleophilic additiona reaction on ketone compound. You can see the reaction…
Q: A student performed the identical experiment to isolate Fe, NaCl, and SiO2 in a mixture. They…
A: Mixtures can be physically separated by using methods that use differences in physical properties to…
Q: )A nickel catalyst is commonly used in the hydrogenation of ethylene. If the initial concentration…
A:
Q: Part C To normalize this wave function, you must calculate the integral ²da. What is the value of…
A:
Q: 11 In the six following sets, which pairs of structural for- mulas represent constitutional isomers?…
A:
Q: 3. In each series circle the most stable and underline the least stable. Explain your choices. a b
A:
Q: What is a key difference between alpha helices and beta sheets? An alpha helix is composed of two or…
A: Alpha-helix and beta-sheets are the types of secondary structure of protein.
Q: (N2H4) is a fuel used by some spacecraft. It is normally oxidized by N204 according to the following…
A: We have been given Standard enthalpy of formation of N2H4 = 50.6KJ/mol Standard enthalpy of…
Q: From the following data, C (graphite) + O₂(g) → CO₂ (g) H₂(g) + O₂(g) → H₂0 (1) CH₂(g) + 20₂(g) →…
A:
Q: Which of the following functions are eigenfunctions of the operator ·Â if  = d² f(x)/dx²? Check all…
A:
Q: Determine the enthalpy of reaction for HCI(g) + NaNO₂ (s) → HNO₂(1) + NaCl(s) 2NaCl(s) + H₂O(1)→…
A:
Q: A 112.3 g piece of copper (specific heat 0.380 J/g °C) is heated and then placed into 400.0 g of…
A:
Q: Ma Nit Ho hồ Phra Ph app.101edu.co Ho Single H Bond H UNO C G wh Multiple Bonds G tar 459 Use the…
A: Thermodynamics is branch of chemistry in which we deal with amount of heat evolved or absorbed by…
Q: It is found that 122 mL of canola oil weighs 112 g. What is the density in grams per milliliter of…
A: Given data, Mass of canola oil = 112 g Volume of canola oil = 122 mL
Q: Find the empirical formula for a compound that has the following percent composition: carbon =…
A: An Empirical formula is the chemical formula of a compound that gives the proportions (ratios) of…
Q: The first-order rate constant for the decomposition of NjOs, given below, at 70°C is 6.82 x 10s.…
A: Answer: Sum of the powers of all concentration terms present in the rate law of the reaction is…
Q: 8. Given the following, what will be the approximate equilibrium pH of an aqueo of ammonium cyanide,…
A: We have find out the solution.
Q: A solution is made by dissolving 0.605 mol of nonelectrolyte solute in 891 g of benzene. Calculate…
A: Answer: When a non-electrolyte, non-volatile solute is added in a solvent it causes a depression in…
Q: What is the IUPAC name for the compound shown? CH3 H3 C-C CH–CH2–CH2CH3 CH3 CH₂ CH3
A:
Q: Express the Beer-Lambert law mathematically and discuss the factors causing the deviation from it.
A: This question belongs to photochemistry chapter.
Q: 19 1. 2. a + KOH Proton transfer b Lewis acid/base c= Radical chain substitution H₂O Submit Answer…
A: There are two types of nucleophilic substitution reaction: Unimolecular nucleophilic substitution…
Q: For the following compound how many different signals would you see in the carbon NMR? (Assume that…
A: -> Number of signal in 13C NMR is equal to number of non equivalent carbon atoms .
Q: The following set of data is described as (assume X,=190): 168, 200, 153, 169 and 162 ppm a.…
A: Accuracy refers to the closeness of a measured value to a standard or known value The closeness of…
Q: Identify the reagent(s) you could use to accomplish the following transformation. Select all that…
A: The given reaction is an example of substitution reaction in which one functional group is replaced…
Q: Draw a resonance structure of the compound shown below, called 2-heptanone, which is found in some…
A:
Q: 4. Identify and label zeaxanthin on the 1:2 plate. Explain the basis for your assignment with refer-…
A: Zeaxanthin is one of the most common Zeaxanthin is one of the most common carotenoids in nature, and…
Q: What is the pH for 5.0 x 10 M Ba(OH), solution? A) 9.0 B) 10.7 C) 9.3 D) 5.0 E) 11.0
A: Given, Concentration of Ba(OH)2 solution = 5.0 × 10-4 M pH of the solution = ? Note: Ba(OH)2 is a…
Q: Images attached show lead solid in aqueous hydrochloric acid. After looking at the images cite…
A: Physical change: In a physical change only the state of matter changes. In this change different…
Q: Complete and balance the following redox equation. What is the coefficient of OH when the equation…
A:
Q: Indicate the signs of ASsys, ASsurr and ASuniv for the complete combustion of propane shown below.…
A: The answer for this problem is provided below in great details within the attached figure.
Q: Macmillan Learning The structure is an anion with three possible resonance contributors. One…
A: Given that, an incomplete resonance structure of a molecule. We have to draw all the resonance…
Q: why do you have -k2 in the L.H.S and -(-w)2 in the R.H.S ?
A:
Q: a. At pH 7, which groups are predominantly deprotonated? none of these groups are depr b. At pH 7,…
A: The deprotonation of the acidic proton depends on the pH of the solution. If the pH of the solution…
Q: 2. Predict the product of the following epoxide opening reactions with correct stereochemistry. a.…
A:
Q: 2. When mixing two types of atoms, say A and B, will atoms always diffuse from regions of higher…
A: Diffusion plays an important role in various biological and chemical processes. Diffusion occurs…
Q: Draw the predominant structure of the tetrapeptide at pH-9 and give the overall charge. You may use…
A: This question is based on structure of peptide.
Q: Question: The absorption spectrum of a dye is shown. What color will the dye appear to be?…
A: •Given:-
Q: Electromagnetic radiation with a wavelength of 525 nm appears as green light to the human eye. The…
A:
Q: The following data are for the gas phase decomposition of sulfuryl chloride at 600 K. SO₂Cl₂(g) […
A:
Q: Provide a name or a structure for each of the following compounds include stereochemistry as…
A:
Q: 8. You are asked to make 100 ml of solution containing the following: Component 1 mM Tris (stock…
A: To calculate the dilution factor, required to prepare (i) 100 mL of 1mM tris solution from 1 M tris…
Q: 59 Which of the following compounds can exist as cis-trans isomers? For each that can, draw both…
A: -> In cis form similar groups are on same side and in trans form similar groups are on opposite…
Q: Calculate the amount of heat needed to melt 113.g of solid ethanol (CH3CH2OH) and bring it to a…
A:
Q: Use the following equation to answer the following question: 3Ba(NO3)2+Al2(SO4)3=3BaSO4+2Al(NO3)3…
A:
Q: In this experiment the mass of NaCl was measured indirectly only. Desgin an experiment procedure…
A: One way to directly measure the mass of NaCl would be to use a technique called gravimetric…
Step by step
Solved in 2 steps with 1 images

- When 0.6403 of g calcium bromide is dissolved in 75.00 mL water, the solution temperature increses by 1.07 °C. Calculate the enthalpy of dissolution (ΔHrxn) for calcium bromide, in kj/mol.(Assume constant pressure is, 41.84j/g. °C as the specific heat capacity of the solution, and a density of 1.00 g/ml). Write out the equation. I am confused on where to start with this problem.Formula: C3H6OMelting Point: -94.0°CBoiling Point: 56.0°CDensity of liquid: 0.791 g/mLHeat of Fusion: 98.14 J/gHeat of vaporization: 538.9 J/gSpecific heat capacity (solid): 1.653 J/g°CSpecific heat capacity (liquid): 2.161 J/g°CSpecific heat capacity (gas): 1.291 J/g°C How much heat is involved in taking 165.2 g of acetone from 14.7°C to 52.1°C?Estimate the thermal properties of a food with the following composition: carbohydrate 0.3%, protein 27.6%, fat 17.5%, ash 4.7%, moisture 49.9%. a. Specific heat is based on the Siebel equation = kJ / kg ° C. b. Specific heat is based on the Charm equation = kJ / kg ° C. c. Specific heat is based on the Heldman & Singh equation = kJ / kg ° C. d. Specific heat is based on the Choi & Okos equation = kJ / kg ° C. e. Heat conductivity is based on the equation Sweat = W / m ° C. f. Heat conductivity is based on the Choi & Okos equation = W / m ° C. g. Heat diffusivity based on the Choi & Okos equation = m² / s.
- A student determines the molar mass of a liquid unknown by the method used in thisexperiment. She found that the equilibrium temperature of a mixture of ice and water was1.0°C on her thermometer. When she added 12.3 g of her unknown sample to the mixture, thetemperature, after thorough stirring, fell to -4.0°C. She then poured off the solution through ascreen into a beaker. The mass of the solution was 93.4 g. Kf = 1.86°C/m What was the molality of the unknown solution? ______________ m How much unknown liquid was in the decanted solution? ______________ gHow much water was in the decanted solution? ______________ gWhat did she find to be the molar mass of the unknown liquid, assuming she made thecalculation properly?______________ gWhen two different liquids are mixed or when a gas or solid is dissolved in a liquid, bondsare broken between neighbouring molecules (or sometimes atom) in the feed materials,and new bonds are formed between neighbouring molecules (or ions) in the productsolution. i. Briefly distinguish between heat of solutions and standard heat of formation of solution.ii. Consider solid X is to be dissolved in solvent Y. If Δ?? °is the heat of formation of X(s),using the appropriate notations, write an equation to calculate the standard heat of formation of solution that contains 5 mol of X in 100 mol of Y at 25°C, relative to X and Y.The specific heat of an unknown liquid is determined using a sample of molybdenum (Mo) with a mass of 35.12g. The Mo is heated to 99.2OC in boiling water, and then transferred to 52.16g of unknown liquid whose temperature is 23.4OC. The final temperature of the Mo-unknown liquid system is 28.9OC. Calculate the specific heat of the unknown liquid. CMo = 0.251 J•g-1•OC-1
- 6.A fuel is burned with 50% excess air and the combustion characteristics of the fuel oil are similar to C12H26. Determine the ff. a) air fuel ratio b) volumetric (molal) analysis of the products of combustion.Table 2. Gibbs Free Energies of formation (kJ), ∆G°f, for Ions in 1M Solution and Ionic Solids Cations Cl--131.228 I--51.57 NO3--108.74 SO4-2-744.53 Ca2+-553.58 -748.1 -528.9 -743.07 -1797.28 W2 ∆G°f of water = -237.129 kJ/mol Calculated values of ∆G°rxn and the ∆Grxn of each box, Predicted results (ppt or no ppt).Observations (Rxn or No Rxn). S or support and R for Refute Cations Cl- I- NO3- SO4-2 Ca+2 help4.38 g of a gas (28.1 g mol–1) was dissolved in liquid water in a calorimeter with a calorimeter constant of 76.0 J K–1. Prior to mixing everything was equilibrated to 21.2 °C. The resulting solution with a mass of 23.4 g was observed to be at a temperature of -6.8 °C and have a specific heat capacity of 4.61 J g–1 K–1. Determine q per mole for this dissolution process. a. -5150 J/mol b. -1.94 × 104 J/mol c. 2.89 × 105 J/mol d. -3.39 × 105 J/mol e. 3.30 × 104 J/mol
- Extintion coeficient for Y is 12801. Calculate the concentration in % by mass of a solution containing 3.50 g of NaCl dissolved in 25000 mg of H2O. 2. Calculate the heat change of the combustion of 1.78 g benzoic acid (MM = 122 g/mol) in a constant bomb calorimeter. As measured, the temperature rises from 15.17°C to 28.52°C and the heat capacity of the bomb plus water is 5.7125 kJ/°C. What is the heat of combustion in kJ/mole?Name two characteristic physical properties that distinguish condensed from gas phases, and one which distinguishes solid from liquid phases. Discuss the differences for each property. Name one colligative property and by an equation (label all variables) and 1-2 sentences the physical meaning. Consider a balloon. What thermodynamic system does it represent? How does this system interact with the environment? Calculate the heat capacity at constant pressure of methane for all its contribution and the total value according to the equipartition theorem. What is a more realistic value at room temperature? Why?



