Do your protons correspond to aldehyde or ketone functionality? Explain why or why not. Are any protons attached to an aromatic ring? Explain why or why not. Are vinylic, allylic, benzylic protons present? Explain why or why not. Are there hydrogens on carbons next to electronegative atoms? Explain why or why not.
Do your protons correspond to aldehyde or ketone functionality? Explain why or why not. Are any protons attached to an aromatic ring? Explain why or why not. Are vinylic, allylic, benzylic protons present? Explain why or why not. Are there hydrogens on carbons next to electronegative atoms? Explain why or why not.
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter13: Gases
Section: Chapter Questions
Problem 117A
Related questions
Question
The following question is from at-home practice material. I have broken the question into 4 parts and the question is attached below. The proton NMR that should be used is attached as well.
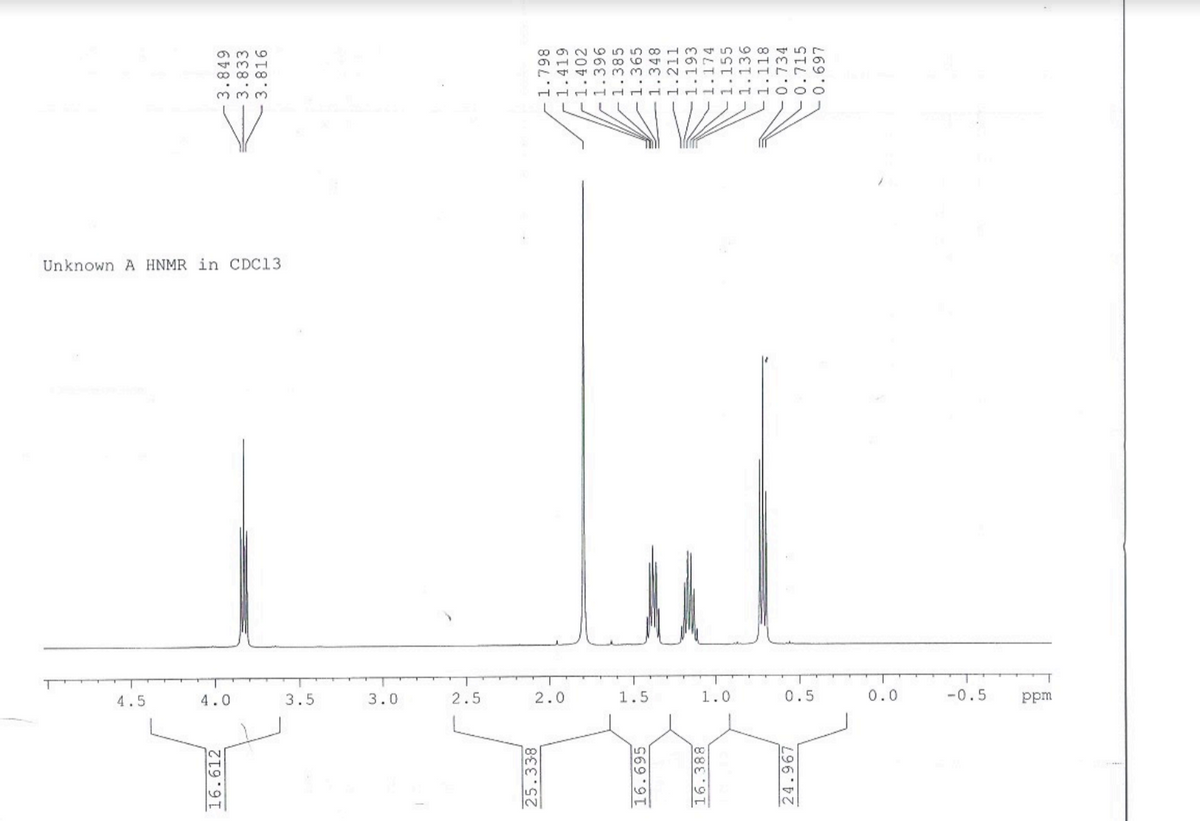
Transcribed Image Text:Unknown A HNMR in CDC13
4.0
3.5
3.0
2.5
2.0
1.5
1.0
0.5
0.0
-0.5
ppm
4.5
16.612
3.849
3.833
3.816
25.338
1.798
1.419
1.402
96
1.385
16.695
1.365
1.348
1.211
1.193
16.388
1.155
1.136
L1.118
-0.734
0.715
24.967
0.697

Transcribed Image Text:Do your protons correspond to aldehyde or ketone functionality? Explain why or why not.
Are
any protons attached to an aromatic ring? Explain why or why not.
Are vinylic, allylic, benzylic protons present? Explain why or why not.
Are there hydrogens on carbons next to electronegative atoms? Explain why or why not.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co