Q: 5 6.90 Nat
A: Henderson equation, pH = pKa + log [salt][acid]
Q: The rate of a certain reaction is given by the following rate law: rate = k[N₂]²[H₂]² Use this…
A: Introduction Reactants are transformed into products at a pace known as the rate of reaction or…
Q: e. He 1. Br2, CH3CO₂H 2. pyridine, heat
A: We can explain this reaction as following shown in next step -
Q: PROBLEM SET (Section 15804 (TR) Functional Groupe: Orde and label the functional groups present CH₂…
A: The given compound is Lysergic acid diethylamide which contains a secondary amine, tertiary amine…
Q: 12) How many grams are there in 1.00 x 1024 molecules of BC13? 13) How many grams are there in 4.50…
A:
Q: The carbons in an alkane are all hybridised >>
A:
Q: Calculate the lowest possible energy for an electron confined in a 1D-box of length equal to 17.8…
A: We know that , the lowest possible energy for an electron :- • En = (n2 h2)/(8mL2)…
Q: 1. Calculate the molar mass of each compound. a. NaNO2 (sodium nitrite), a preservative in hot dogs,…
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: Hi, I'm learning about Density in chemistry , and I'm confused about the exemplos that the professor…
A: The significant figures in an operation is found out based on the number of significant figure…
Q: At the end of this, how did we go from a 5 to 1 ratio moles to 2CO2 and 3H2O?
A:
Q: How many orbitals have n = 3 and m? = -1?
A: Principal quantum number: It is represented with letter . The allowed n values are all positive…
Q: 1. How many moles of He are there in a sample that contains 13.4 grams of hellum? moles 2. How many…
A: The relationship between moles and mass is: Moles =Given massMolar mass ......(1)
Q: a. disulfur tetrafluoride b. carbon trioxide c. nitrogen pentoxide I d. nitrogen tribromide e.…
A: Given in following question many compounds give molecular formula for each compound these compounds…
Q: Question 3 A chemist accidentally leaves a 300.0 g sample of benzene on an activated hotplate. The…
A: Given Mass of Sample (m) = 300 g = 3001000=0.3 kg Initial temperature (t1) = 250C= (25+273)K=298 K…
Q: A graduated cylinder which has been filled with 20.00 mL of water. A solid object is added to the…
A: Density of a substance defined as" It is the ratio of mass of a substance to its volume " Density =…
Q: Does the following molecule obey Hückel’s rule? Show the calculations
A: According to the Huckel rule, a molecule will be aromatic if it contains 4n+2 π electrons, here…
Q: kelvins (K) appear in regular type, while those for variables such as mass (m), volume (V), and time…
A:
Q: Is-N(CH3)2 an o/p- or a m-directing agent during SEAR? Draw the o- and m- resonance structures to…
A: In electrophilic aromatic substitution reaction an atom that is attached to an aromatic system is…
Q: A hypothetical element has 3 stable isotopes. The masses of the isotopes and percent abundance of…
A: The percentage abundance is the amount of isotope in nature. The higher the percentage abundance,…
Q: Based upon your knowledge of mordants, which could be used as a of the following compounds mordant?
A: Mordant is a type of element which can form coordination complex and these complex helps to dye in…
Q: 2 d out of A student's house contains 400kg of air. In winter the student turns down the heat as…
A: Solutions- Weight of Air = 400kg T1 = 14°C = 287K, T2 = 21°C = 294K Find - Energy in kJ
Q: (a) Below are the amino acids aspartic acid (Asp), tyrosine (Tyr), arginine (Arg), and serine (Ser).…
A: here we are required to draw given tetrapeptide at neutral pH and also need to identify the N…
Q: НО + Br₂ AlBr3
A: Benzene and it's derivatives undergo electrophilic substitution reactions in which incoming…
Q: 6. At a certain temperature, the reaction CO (g) + Cl₂ (g) = COCl₂ (g), has an equilibrium constant…
A:
Q: An equation for a double replacement reaction is a) Fe) + CuSO4(aq) → FeSO4 (aq) + Cu(s) b) 2 K+ 2…
A:
Q: Practice Example A: With as much detail as possible, describe the phase changes that would occur if…
A: Given: Phase diagram of water To find: the phase changes occurring from point R to point P and then…
Q: Calculate the weight percent of ascorbic acid in a tablet of Vitamin C using correct significant…
A:
Q: Which of the following functional groups is NOT found in amino acids? A. amine B. amide C.…
A: Given options of functional groups are A. amine B. amide C. carboxyl D. aldehyde
Q: a. How many valence electrons does Sn have, and what are the specific valence electrons for Sn?…
A: Valence electrons are those electrons of an atom that are present in the outermost shell of the…
Q: Dinitrogen tetroxide, N204, and hydrazine, N2H4, react explosively to produce nitrogen gas, N2, and…
A: Given balanced equation: N2O4 + 2N2H4 --> 3 N2 + 4 H2O Calculate the moles of N2O4. mass of…
Q: 15. When solid lithium metal is exposed to pure nitrogen gas, a rapid reaction occurs. The only…
A: Lithium has high reactivity towards nitrogen and it rapidly reacts with N2 to form lithium nitride.
Q: Ph H3C How would you draw the Molecular Orbita Diagram for this radical?
A:
Q: Write the balanced molecular equations (include all counter ions) for the confirmation step of each…
A:
Q: H :O: || H-C-C-H Н. molecule 1 H H molecule 2 :0: H HH H conjugate acid of molecule 1 Click and drag…
A:
Q: Which chemical equation is consistent with the concentration vs. time data below? Circle your answer…
A:
Q: Acetyl chloride test must be performed in dry test tube otherwise the unknown will give a false…
A: In acetyl chloride test, the unknown is first dried by using anhydrous calcium sulfate to remove any…
Q: CuO + 1. Reaction 4: a. Color of copper(II) sulfate b. Water solubility of copper(II) sulfate 2.…
A: Copper one of the most well-known metallic chemical elements for heat and electricity conductivity,…
Q: 3.2(a) Calculate the molar entropy of a constant-volume sample of neon at 500 K given that it is…
A:
Q: A solution contains 0.84 M (mol/L) of AICI3. What is the ionic strength of the solution in mol/L?…
A:
Q: Question 5 The molecule 12C¹60 has a moment of inertia 1.448 x 10-46 kg m³. For what value of the…
A: Given: moment of inertia, I = 1.448 × 10-46 kgm3 Temperature= 300K Rotational energy closest to the…
Q: Draw the structure that represents the compound C8H9Br HNMR: 7.4 (doublet, 2H), 7.2 (doublet, 2H),…
A: We have to determine the structure of the compound C8H9Br using the given NMR data
Q: 4. Predict How will an increase in pressure affect the following chemical equilib- rum: 2NOCI (gas)…
A: According to Le Chatelier's principle temperature, pressure , concentration, inert gas etc. will…
Q: Use the References to access important values if needed for this question. When aqueous solutions of…
A:
Q: What class of mineral forms when element C is bonded with element O, as in the mineral calcite? 29…
A: Calcite is calcium carbonate. The formula of calcite is CaCO3 .
Q: oxidation-reduction reactions. Show all steps in the half the oxidizing agent and reducing agent.…
A: Given Oxidation-reduction reactions a. FeCl2 + KMnO4 + HCl →FeCl3 + KCl + MnCl2 + H2O And b.…
Q: 1 For a particle in a one dimensional square well in which the potential energy rises to a finite…
A: For a particle in a one dimensional square well in which the potential energy rises to a finite…
Q: In a density measurement, you determine the mass of an object to be 10.50 +/- 0.02 g and the volume…
A: The error in a measurement is calculated by the real value of measurement and error in the component…
Q: aw the Lewis structure created by the curved arrow. Write in formal charges wherever they are not…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: Prepare 100 plates of Mueller Hinton Agar using 100mm petri dish. (Manufacturer's instructions:…
A: Total number of plates of Mueller Hinton Agar to be prepared = 100 Volume of agar solution in each…
Q: study of the gas-phase oxidation of nitrogen monoxide at 25°C and 1.00 atm pressure gave the…
A: We have to first determine the coefficient from the experiment given in the question. Using…
Does the following molecule obey
Show the calculations.
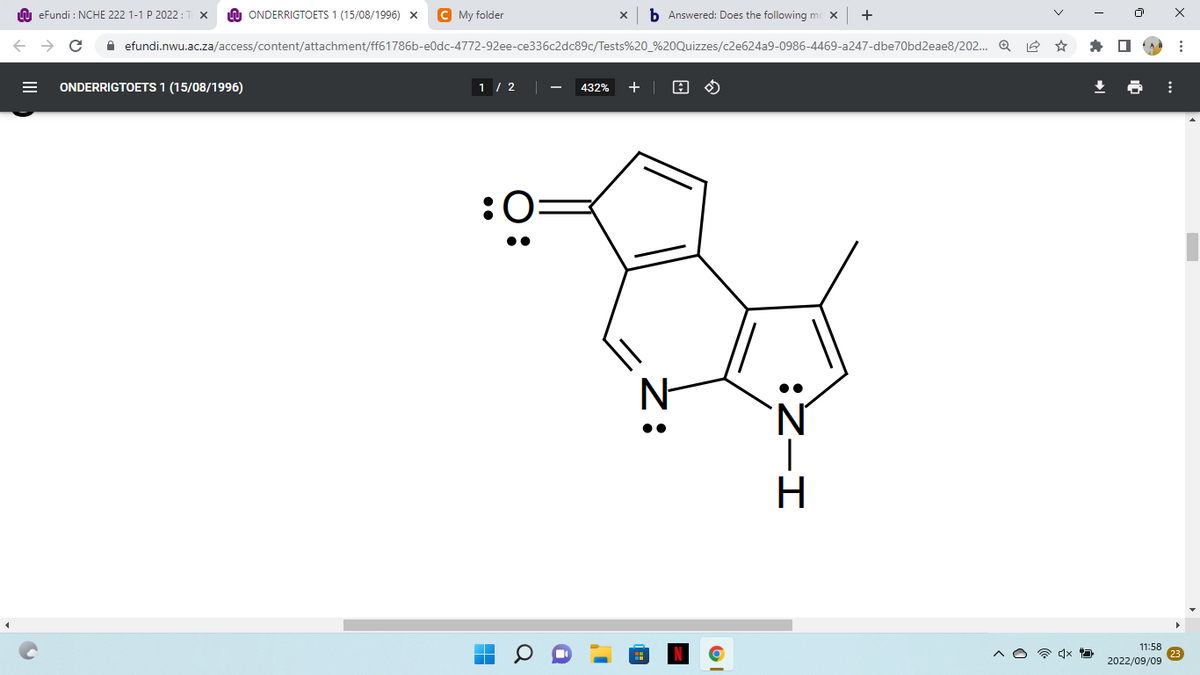
Step by step
Solved in 2 steps

- 10.11/ A 1.5-kg specimen of a 90 wt% Pb-10 wt% Sn alloy is heated to 250°C (480°F), at which temperature it is entirely an a-phase solid solution . The alloy is to be melted to the extent that 50% of the speci- men is liquid, the remainder being the a phase. This may be accomplished either by heating the alloy or changing its composi- tion while holding the temperature con- stant. (a) To what temperature must the speci- men be heated? (b) How much tin must be added to the 1.5-kg specimen at 250°C to achieve this state? 10.15) A magnesium-lead alloy of mass 5.5 ka consists of a solid a phase that has a compo- sition that is just slightly below the solubility limit at 200°C (390°F). (a) What mass of lead is in the alloy? (b) If the alloy is heated to 350°C (660°F). how much more lead may be dissolved in the a phase without exceeding the solubility limit of this phase? 10.27 A hypothetical A-B alloy of composition 55 wt% B-45 wt% A at some temperature is found to consist of mass…image https://cxp.cengage.com/contentservice/assets/owms01h/references/chemtables/gen_chem/eo.html ^ tablehttps://m.youtube.com/watch?v=vM1SP346XBc&list=PLeJOSNLNZfHubfLdq0kOayASeUllMOGn4&index=4 I watched this the lecture video over and over and I am allowed to work with someone but I am having trouble with part B and I provided the YouTube link of the data or video attached to this lab
- 1-Pentanol to 1-bromopentane Chemicals: - 60ml Conc. Sulfuric Acid - 100ml Saturated Sodium bicarbonate - 65ml 1-Pentanol - 78g sodium bromide - Distilled water - 58.42g 1-Bromopentane 1-Pentanol Sodium Bromide Sulfuric Acid 1-Bromopentane Formula C5H12O NaBr H2SO4 C5H11Br MW (g/mol) 88.15 102.894 98.078 151.04 Density (g/mL) 0.811 3.21 1.84 1.218 Boiling point (*C) 138 1,396 337 130 NaBr(aq) + H2SO4(aq) -> NaHSO4(aq) + HBr(aq) CH3(CH2)4OH(aq) + H+ Br- (aq) CH3(CH2)4OH2 (aq) + Br-(aq) CH3(CH2)4OH2 (aq) + Br-(aq) CH3(CH2)4Br(aq) + H2O(aq) How do I calculate the percent yield and identify the limiting reagent?Chloroform has ∆Hvaporization = 29.2 ??/??? and boils at 61.2 °C. What is the value of ∆Svaporization for chloroform?Plz o Asap...!
- There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…How to convert 4.76pm to um? Showing the work. 4.76pm x___________m x_____________ um =________________um How to convert 25.0mL to cL? Showing the work. 25.0mL x______________ x ___________ = ______________cL



