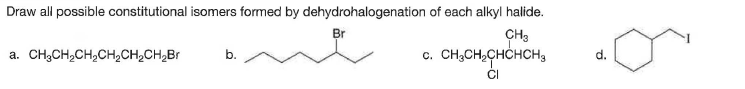
Draw all possible constitutional isomers formed by dehydrohalogenation of each alkyl halide. Br CH3 c. CH,CH,CHCHCH3 a. CH3CH,CH,CH2CH¿CH,Br b. d. CI
Q: Draw the products in each reaction. CH,CH3 CH;CH,OH K* -OC(CH,)3 а. Он CH3CH2O b. CH3-C-CH,CH3 d. CI
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: CHO [C2]= accomplish the synthetic transformations using any reagents containing NO MORE THAN 2…
A: A Grignard reagent is vastly used in organic synthesis to introduce a new C-C bond in a molecule.…
Q: Which one of the following is the best(faster reaction) nucleophile for the 2-bromobutane SN1…
A: the strength of nucleophile does not affect the reaction rate of SN1 because the nucleophile is not…
Q: H20 `CI
A: It is a tertiary alkyl halide. Hence, both SN^1 and E1 reaction will be favoured. But, water is a…
Q: Draw the product formed by treating each compound with excess CH3I, followed by Ag2O, and then heat.
A: The complete reaction for a is shown below.
Q: Draw the organic product of each reaction. a. Br "OH + "OCH(CH3)2 С. b. + "OCH3 d. Br + "OCH,CH3
A: The reactions given are,
Q: Be sure to answer all parts. Draw a stepwise mechanism for the following reaction: HO H,SO, Part 1:…
A: This reaction is dehydration reaction which means water leaves out the reactant. Under acidic…
Q: 1. Which alkene is the major product of this dehydration? HO H2SO4 heat А В C D
A:
Q: Draw a stepwise mechanism for each reaction. Br + CH3CH2NH2 NaOH 'N" + H20 + NaBr a. Br ČH,CH3 NABH4…
A: When aldehyde or ketone reacts with NaBH4, they get reduced to alcohol. Cyclization is defined as…
Q: Draw the product of each reaction. wCH3 HBr CH3CH,OH c. CH;CH2 ČHĄCH, a. H2SO4 CH [1]-CN [1] CH30 b.…
A: In acidic conditions, an epoxide opens from more substituted side to give the corresponding product.…
Q: Draw the major organic product for each synthetic reaction step. CH;CH2CI HNO3 2 mol. Br2 Product A…
A: Friedel-Craft alkylation reaction: When the benzene reacts with an alkyl halide in presence of Lewis…
Q: Reagents 1. Li(CH3)2Cu 2. H30* 1. Li(CgHs)2Cu 2. H30* 1. Li(CH2=CH)2Cu 2. H30* a i 1. NaBH4 f CH212…
A: This question is demanding the basic knowledge of organic reagents which can be used for the…
Q: What alkene can be used to prepare each alcohol as the exclusive product of a two-step…
A: The reaction in which boron and hydrogen is attached to the alkene and formed alcohol is known as…
Q: What alkane is needed to make each alkyl halide by radical halogenation?
A:
Q: What reagents are required to convert tert-butylbenzene to p-tert- butylbromobenzene? A) HBr Br B)…
A: THIS IS AN ELECTROPHILIC SUBSTITUTION RACTION OF THE BENZENE RING,WHERE Br2 IS THE ELECTROPHILE.
Q: What alkenes are formed from each alkyl halide by an E1 reaction? Use the Zaitsev rule to predict…
A: Zaitsev rule: During elimination greater substituted alkene is predominantly formed.
Q: Draw the most stable form of the major mixed Claisen product formed in the reaction. Select Draw…
A: Claisen condensation is the reaction in which enolates are added to an ester molecule. Enolate is…
Q: These reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O,…
A:
Q: B- Instructions: What reagent and/or catalysts are necessary to bring about each conversion? Br 1.…
A:
Q: What alkene is the major product formed from each alkyl halide in an E1 reaction?
A: E1 reactions are two step reactions in which the first step is the cleavage of carbon halogen bond…
Q: 30. Draw all of E2 elimination product with the correct stereochemistry. Label the major product. Br…
A: Following is the major product of the given chemical reaction.
Q: Which alkyl halide will react the fastest in an SN2 reaction (CH3)3CI CH3CH2Br CH3CH2I CH3I CH3Br
A: The SN2 reactions is used to denote bimolecular nucleophilic substitution. In this mechanism, a…
Q: Label the α and β carbons in each alkyl halide. Draw all possible elimination products formed when…
A: The carbon to which the functional group is attached is called the alpha carbon. The next carbon to…
Q: Br "OCH,CH3
A: Reaction of alkyl halides with nucleophile is termed as nucleophilic substitution. It is competed by…
Q: Identify the missing reagent: 1) ???? 2) NaCN OH CN O PB13 O TSCI (Ts: Tosyl) O CISIME3 (Trimethyl…
A: PBr3 can not be used because it's give SN2 reaction so that configuration inverse and then in second…
Q: Draw the major organic product of the following reaction conditions. CH3 1. OsO4, H20 2. NalO4 ČH3
A: Organic reactions
Q: What alkenes are formed from each alkyl halide by an E2 reaction? Use the Zaitsev rule to predict…
A: Given compounds,
Q: Select the alkyl halides which reacts fastest in an SN1 reaction. Br A. CH3 CH₂CI B. C. D. CH3 CH₂…
A: We have to determine the alkyl halide that will react fastest
Q: H;C-CH- -CH2- -CH2 CH3 CH3 Common Name 5. What resources could potentially be used to stabilize a…
A: Common name :- Propyl isobutyrate
Q: Which reagents lead to this transformation? (A) (B) (C) (D) ??? O₂N. 1. Br₂, FeBr3 2. Mg 3. CH,CHO…
A: This question is based on the Electrophilic aromatic substitution reaction.
Q: What alkenes are formed from each alkyl halide by an E1 reaction? Use the Zaitsev rule to predict…
A: By using the Zaitsev's rule the major product can be predicted as it states that the more…
Q: What is the most proper name of the following ether? OCH3 a. (25,3R)-2-methoxy-3-methylpentane b.…
A: Write proper name of the given ether ?
Q: Do ethers have a good leaving group ?
A: Ethers are the organic compounds which have the general formula R-O-R'. A group which leaves as a…
Q: R OR1 -R2 R R-CEN
A:
Q: Draw the substitution and elimination products.
A: Hey since you have posted a question with multiple sub-parts, we will solve first three sub-parts…
Q: What is the most proper name of the following ether? OCH3 a. (25,3R)-2-methoxy-3-methylpentane b.…
A: The IUPAC name of the following ether is
Q: Label the α and β carbons in attached alkyl halide. Draw all possible elimination products formed…
A: Dehydrohalogenation Reaction: The secondary halide undergo either elimination or SN2 depending upon…
Q: What reagents do you need to use to for the following reactions? Match the letter with the reagent!…
A: Given conversions are epoxide ring formation from alkene and epoxide ring opening by using…
Q: Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2
A: Introduction: NaNH2 is a strong base. It is often used for deprotonation in weak acids or also for…
Q: 10. Assigh E or Z configuration for each alkene shown below. EN H3CH2C. `CH2NH2 H3C HO CI
A: According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority. 2) If first atom…
Q: Draw the alkynes formed when each dihalide is treated with excess base.
A: a) When the given compound (1,2-dichlorobutyl)cyclohexane is treated with an excess base such as…
Q: Which halide is most reactive in SNl reactions? Select one: O H;C-CH2-CH2CI CH3 H3C-CH-CH2CI CH3…
A:
Q: ? Br What is the best reagent for the transformation shown above? B E „Mgl A D PPH3
A: We know that R-Li gives exclusively gives 1,2 product.
Q: Draw the products formed when each dihalide is treated with excess NaNH2.
A: a. The final product will be alkene if monohalide undergoes elimination, however, the final product…
Q: Draw the major organic product. H,C CH, Br H H3C CH3 H2 HC CH H3C- H H-Br H
A: HBr addition in alkene proceeds via formation of a stable carbocation. The mechanism is shown below…
Q: What are the products of this NBS bromination reaction? CD2=CH-CH3 + N-Br hv (A) CHD=CD-CH,-Br and…
A: The answer is given as follows
Q: Which halide is most reactive in SN2 reactions? Select one: CH3 H3C-C-CH2CI CH3 CH3 H3C-CH-CI O…
A: Which halide is most reactive in SN2 reation?
Q: What is the product of the following reaction? H3C-CH,-CH=CH2 HBr JF-MO5 H;C-CH-CH2-CH3 Br JF-MO5а…
A: Anti Markovnikov rule portrays the regiochemistry in which the substituent is clung to a less subbed…
Q: 7. For each reaction, circle the correct product. Br N2OCH3 or NaOCH3 or E2 E2
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: Draw the major product of this reaction: HgSO4 H3C =CH3
A: This reaction is the addition of catalytical hydration on alkyne Key points form final product…
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images

- What other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X if no carbocation rearrangement occurred?Which alkene in each pair is more stable?(a) What product(s) are formed when the E isomer of C6H5CH = CHC6H5 is treated with Br2, followed by one equivalent of KOH? Label the resulting alkene(s) as E or Z. (b) What product(s) are formed when the Z isomer of C6H5CH = CHC6H5 is subjected to the same reaction sequence? (c) How are the compounds in parts (a) and (b) related to each other?
- The 1,2‑dibromide is synthesized from an alkene starting material. Draw the alkene starting material. Clearly, show stereochemistry of the alkene.Rank the species in each group in order of increasing nucleophilicity.a. CH3CH2S-, CH3CH2O-, CH3CO2- in CH3OHb. CH3NH2, CH3SH, CH3OH in acetonec. -OH, F-, Cl- in acetoned. HS-, F-, Cl- in CH3OHcomplete the conversion using SN1, SN2, E1, or E2. what is a possible reagent? cyclohexane --> cyclodi-1, 3- ene
- Which group in each pair is assigned the higher priority? a. – CH3, – CH2CH3 b. – I, – Br c. – H, – D d. – CH2Br, – CH2CH2Br e. – CH2CH2Cl, – CH2CH(CH3)2 f. – CH2OH, – CHODraw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.Replacing a kin, a word each of the following our kids results in a significantly lower reaction. Identify the reasons for this decrease in rate for each alkene D – F. Select all that apply. 

