Q: Q3. Name the following compounds according to IUPAC rules: CI CI CI F
A: Since you have posted a question with multiple sub-parts, we will solve first three subpartsfor you.…
Q: How many π electrons are contained in each molecule?
A: a. We first check for the total noof double bonds which is 3 and one electron is a radical. There…
Q: Draw three additional resonance structures that demonstrate which carbon atoms are more electron…
A:
Q: Part A H;C CH-N-CH, + HBr(aq) ? H3C Draw the molecule on the canvas by choosing b toolbars. The…
A: Ans
Q: B1) Draw all the Resonance structures for Cycloheptatrienyl anion, using arrows show the flow of…
A:
Q: RULES O Lowest number to priority O First Point of Difference O Alphabetization as tiebreaker Parent…
A: Since you have asked multiple questions, we will solve the first one for you, for remaining ones…
Q: Draw Kekulé’s structureof benzene, and explain why it does not truly representthe actual structure.
A: Kekule gives the structure of benzene that is 6 membered ring of carbon atom with alternate double…
Q: how to draw at least one or two structures from the molecular formula after calculating DU?
A: DU = Degree of unsaturation
Q: How many hydrogen atoms are connected to the indicated carbon atom? a
A: A carbon atom completes its octet only by sharing its valence electrons with other atoms. As a…
Q: of c. H3C-CH-CH-CH2-c-NH-CH2-CH3 IUPAC H3C-CH-CH-CH2-C-NH-CH2-CH3 Common d. H3C-CH2-CH2 -C- -CH3…
A: All the organic compounds are made of a hydrocarbon chain with one or more functional groups…
Q: The chemical structure of ethanol (CH,CH,OH) is shown below. Highlight each atom that is in a methyl…
A:
Q: CI. Molecular formula
A:
Q: Part A Draw the E isomer for CH3CH2C=CHCH2CH3 1. Cl Draw the molecule on the canvas by choosing…
A: When theses are three or four groups attached to the carbon atoms of double bond, it becomes…
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A:
Q: DIRECTIONS:Provide the required number of isomers ner molecular formula by illustrating them in…
A:
Q: - Which of the following choices is correct for diphenyl? Number Number Number Number of of pi of of…
A: option-c is the correct choice
Q: Determine the correct first curved arrow movement of electrons in the following reaction. OH O A)…
A: In the question, it has been asked to find a possible reaction mechanism for the reaction that will…
Q: Indicate the number of bonds and lone pairs of electrons on each of the following atoms: (a) a…
A:
Q: HO.
A: The given compound contains two functional groups such as -OH and olefinic bond. The isomers can be…
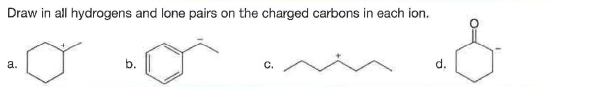
Q: Draw in all hydrogens and lone pairs on the charged carbons in each ion ?
A: A molecule in which carbon has a positive charge along with usually three groups attached is known…
Q: geometry of 1-propanol on each carbon and the oxygen? include a drawing
A: Propanol is C3H7OH and its structure is:
Q: A) For what condition did the FDA approve this drug? B) How many aromatic rings does this drug…
A: A ring is considered to be an aromatic if it is cyclic, planar, have delocalised pi electrons and…
Q: Match correctly all functional group for the lettered molecules: . А с E OH NH₂ G в D F ОН
A: Here we have to write the name of functional group present in the following given compounds.
Q: e of hydrocarbon ne Molecular Formula Condensed formula Structural formula
A: Alkyne can be defined as a hydrocarbon that contain triple bonds between carbon atoms in an organic…
Q: are these aromatic, antiaromatic, or nonaromatic? Draw in all lonepairs and indicate the orbital…
A: for molecule have aromatic, the compound must be cyclic and planner and follow the huckel rule the…
Q: a b CH CH₂ CH₂ CH₂
A:
Q: Draw both a bond-line and fully condensed structure (do show double bond in condensed) for allyl…
A: Fully condensed structure are the representation which show all the atoms but vertical bonds are…
Q: ne resonance hybrid a
A: Rules to identify the best resonance contributors are as follows: Greatest number of full octets…
Q: (a) Add curved arrows to show how the starting material A is converted to the product B. (b) Draw…
A: Given, the structure of A is: The curved arrows to convert A into B is shown below:
Q: (a) Label each carbon, oxygen, and nitrogen atom as being sp, sp, or sp hybridized. (b) Circle and…
A: In a carbon compounds the single bonded carbon atoms possess sp3, double bonded carbon atoms possess…
Q: H2N Refer to the given structure, between group A and B which has higher priority based on…
A:
Q: Name the following structures. (a) (b) (c) CI ÇI
A: (a) The given structure is, The parent carbon chain is hexane. In the second carbon atom, a triple…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided resonance structures,…
A:
Q: H C-H н—с—о- -C- -0-H H
A: 1. It's a common method for representing species attached with carbon atom. 2. Wedge and dash…
Q: the "draw structure" buttor the structure of a compounc ster of molecular formula C
A: Organic compounds can be defined as the compounds that contain carbon and hydrogen atoms in a…
Q: what are the fncional groups presenm MAspirn which has the folbwing structural formula -CH3
A: Aspirine molecule is an aromatic compound containing benzene ring with two substituents on two…
Q: Draw a condensed structural formal for the following molecules: a) b) он
A:
Q: Part B Benzene has a 6-carbon ring. with alternating single and double bonds. Consider a benzene…
A: The benzene is an aromatic hydrocarbon that contains a six-carbon atom ring with an alternate single…
Q: (a) Label each carbon, oxygen, and nitrogen atom as being sp, sp?, or sp hybridized. (b) Circle and…
A: Single bonded carbons possess sp3, double bonded carbon possess sp2 and triple bonded carbons are…
Q: Draw the Lewis structure of OF,. Include lone pairs on all atoms, where appropriate. Select Draw…
A: Electronic configuration: O : 2,6 F : 2,7
Q: CI Cl2
A:
Q: Organic Compounds Part A: Building Models Functional group,# val e's, lewis structure, Molecular…
A: Solution In chemical science, a practical cluster could be a substituent or moiety in an exceedingly…
Q: Whats the name of step and 1 and drar mechanksm arrows even showing electrons on oxygen atoms
A: The name of step 1 is radical initiation as we can see that there is the generation of the radicals…
Q: I dont understand these 2 problems!!!!!
A: In the molecule of cyanazine C, O, H and Cl atom are present.In the outer shell of C atom four…
Q: draw all hydrogen atoms explicitly 2,4-diboguso-1-propylbenzene see Bg on attached special…
A: A systematic structure can be drawn for an organic compound from its IUPAC nomenclature. Write the…
Q: Convert each skeletal structure to a complete structure with all atoms drawn in. a. edit…
A:
Q: Bond-line Formula IUPAC Name a. b. d.
A: 1.Choose largest carbon chain . 2. Start numbering so that sum of substituted group has less numbers…
Q: How many hydrogens are present in the following molecules? a) b)
A: The number of hydrogen molecules in the given structure has to be given,
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Whats the name of step and 1 and drar mechanksm arrows even showing electrons on oxygen atomsA. Draw the meantime r of ephedrine. You can use line structures or a hybrid line structure/condense drawing like the one shown. Be sure it is clear which bonds are wedge and which are dash. B. Draw a diastereomer of ephedrine.Draw the molecule(s)
- Draw the molecule with bonds and and single bonds if neededIn HF , neither H nor F holds a full formal charge of +1 or 1 . Organic chemists represent apartial charge using the Greek letter delta () . On the electron density map of the molecule HF above, add a + to one atom and a to the other to indicate which way the bond is polarized.how to draw at least one or two structures from the molecular formula after calculating DU?
- Show the stepwise mechanism. Show all lone pairs and formal chargesQ2 Show and state the alkyl group that is present in each species(a) Add curved arrows to show how the starting material A is converted to the product B. (b) Draw all reasonable resonance structures for B. (c) Draw the resonance hybrid for B.
- I finished the worksheet but I'm a bit confused when it asks about the DEPT. I also want to make sure the # of carbon groups are correct.See the Attachment & solev the followings (a) Add curved arrows to show how the starting material A is convertedto the product B. (b) Draw all reasonable resonance structures for B. (c) Draw the resonance hybrid for B.Which line (A or B) corresponds to the energy change for an ortho/para substitution?

