Draw the major organic substitution product(s) for (2R,3S)-2-bromo-3-methylpentane reacting with the given nucleophile. İndicate the stereochemistry, including H's, at each stereogenic center. Omit any byproducts. Н Br CH3CH2O (conc.)
Draw the major organic substitution product(s) for (2R,3S)-2-bromo-3-methylpentane reacting with the given nucleophile. İndicate the stereochemistry, including H's, at each stereogenic center. Omit any byproducts. Н Br CH3CH2O (conc.)
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 30MP: The carbocation electrophile in a Friede1-Crafts reaction can be generated by an alternate means...
Related questions
Question
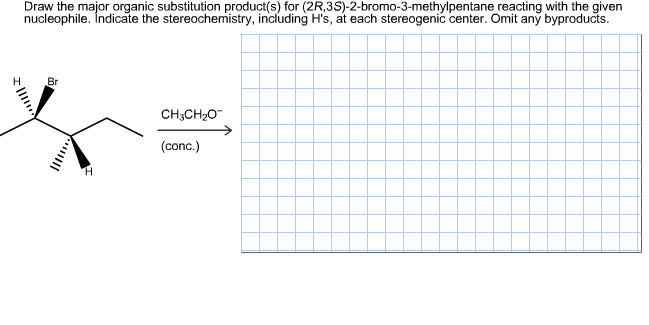
Transcribed Image Text:Draw the major organic substitution product(s) for (2R,3S)-2-bromo-3-methylpentane reacting with the given
nucleophile. İndicate the stereochemistry, including H's, at each stereogenic center. Omit any byproducts.
Н
Br
CH3CH2O
(conc.)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning