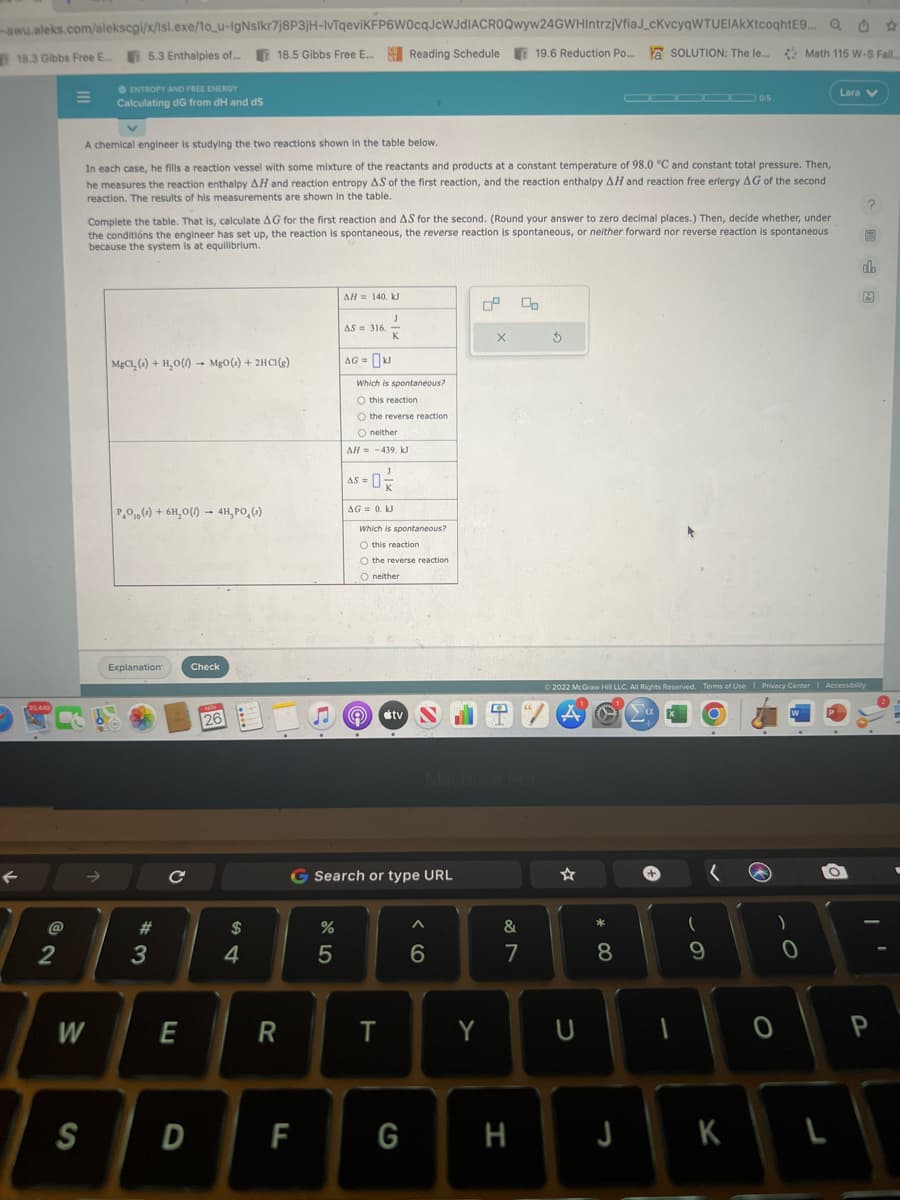
E ENTROPY AND FREE ENERGY Calculating dG from dH and dS A chemical engineer is studying the two reactions shown in the table below. In each case, he fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 98.0 °C and constant total pressure. Then, he measures the reaction enthalpy AH and reaction entropy AS of the first reaction, and the reaction enthalpy AH and reaction free erfergy AG of the second reaction. The results of his measurements are shown in the table. Complete the table. That is, calculate AG for the first reaction and AS for the second. (Round your answer to zero decimal places.) Then, decide whether, under the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward nor reverse reaction is spontaneous because the system is at equilibrium. MgCl, ()+ H₂O()- MgO() + 2HC1(e) PO () + 6H₂O() - 4H, PO () Explanation Check 26 **** AH = 140, KJ AS = 316. AG= Which is spontaneous? O this reaction O the reverse reaction O neither AH = -439, KJ K AS = -0-/22 AG = 0. KJ Which is spontaneous? O this reaction O the reverse reaction O neither (a tv 2 X Lara V Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibility A @ So
E ENTROPY AND FREE ENERGY Calculating dG from dH and dS A chemical engineer is studying the two reactions shown in the table below. In each case, he fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 98.0 °C and constant total pressure. Then, he measures the reaction enthalpy AH and reaction entropy AS of the first reaction, and the reaction enthalpy AH and reaction free erfergy AG of the second reaction. The results of his measurements are shown in the table. Complete the table. That is, calculate AG for the first reaction and AS for the second. (Round your answer to zero decimal places.) Then, decide whether, under the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward nor reverse reaction is spontaneous because the system is at equilibrium. MgCl, ()+ H₂O()- MgO() + 2HC1(e) PO () + 6H₂O() - 4H, PO () Explanation Check 26 **** AH = 140, KJ AS = 316. AG= Which is spontaneous? O this reaction O the reverse reaction O neither AH = -439, KJ K AS = -0-/22 AG = 0. KJ Which is spontaneous? O this reaction O the reverse reaction O neither (a tv 2 X Lara V Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibility A @ So
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter17: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 128IP: Some nonelectrolyte solute (molar mass = 142 g/mol) was dissolved in 150. mL of a solvent (density =...
Related questions
Question
Transcribed Image Text:Q
awu.aleks.com/alekscgi/x/isl.exe/1o_u-IgNsikr7j8P3jH-IvTqeviKFP6W0cqJcWJdIACROQwyw24GWHIntrzjVfiaJ_cKvcyqWTUEIAKXtcoqhtE9...
Gibbs Free E... Reading Schedule 19.6 Reduction Po... Y SOLUTION: The le... Math 115 W-S Fall..
18.3 Gibbs Free E... 5.3 Enthalpies of...
5.3 Enthalpies of... 18.5 Gibbs Free E...
18.5
20,449
2
E
W
S
ENTROPY AND FREE ENERGY
Calculating dG from dH and dS
A chemical engineer is studying the two reactions shown in the table below.
In each case, he fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 98.0 °C and constant total pressure. Then,
he measures the reaction enthalpy AH and reaction entropy AS of the first reaction, and the reaction enthalpy AH and reaction free erfergy AG of the second
reaction. The results of his measurements are shown in the table.
Complete the table. That is, calculate AG for the first reaction and AS for the second. (Round your answer to zero decimal places.) Then, decide whether, under
the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward nor reverse reaction is spontaneous
because the system is at equilibrium.
MgCl, (s) + H₂O() MgO(s) + 2HC1(e)
PO () + 6H₂O(1) 4H, PO ()
Explanation
#3
с
E
D
Check
NOV
26
$
4
R
.
F
AH 140, kJ
%
5
AS 316.
AG = KJ
J
K
Which is spontaneous?
O this reaction
O the reverse reaction.
O neither
AH = -439. kJ
J
AS = 0²/2
-0₁
AG= 0. kJ
Which is spontaneous?
O this reaction
O the reverse reaction
O neither
.
G Search or type URL
T
tv N
6
Y
X
Do
&
7
H
Ś
O▬▬OD 0/5
U
* 00
8
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center I Accessibility
"/A
0
0
1
(
9
JK
)
0
W
Lara V
0
?
db
A
P
T
L
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,