Each diastereomer, if treated with sodium ethoxide (NaOCH2CH3), undergoes an E2 reaction. One of the diastereomers undergoes an E2 to make a single compound. The other diastereomers undergoes E2 eliminations leading to two different compounds. Draw the mechanisms for all three of these E2 reactions, being sure to use conformational structures for the substrate in each reaction. Which of the two diastereomers will undergo an E2 reaction at the faster rate? Why? (In organic chemistry it is often said one picture is worth a thousand words. Be sure to include a picture that supports whatever argument you are making.
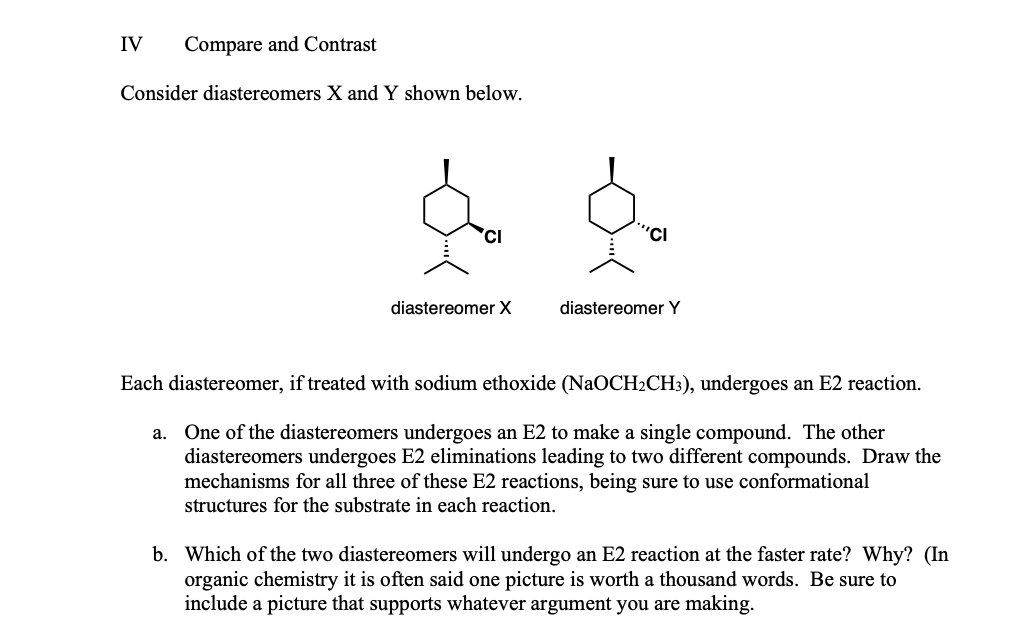
Consider diastereomers X and Y shown below.
Cl
diastereomer X
Cl
HO
H
OH
H
Each diastereomer, if treated with sodium ethoxide (NaOCH2CH3), undergoes an E2 reaction.
-
One of the diastereomers undergoes an E2 to make a single compound. The other diastereomers undergoes E2 eliminations leading to two different compounds. Draw the mechanisms for all three of these E2 reactions, being sure to use conformational structures for the substrate in each reaction.
-
Which of the two diastereomers will undergo an E2 reaction at the faster rate? Why? (In
organic chemistry it is often said one picture is worth a thousand words. Be sure to include a picture that supports whatever argument you are making.
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images






