Eight replicate measurements were made on a test solution producing the following results for the concentration of sodium ion (mg/L): 63.91, 65.92, 69.98, 64.24, 65.45, 65.82, 66.51, 65.11. The value 69.98 is an outlier suspect. A Q-test at 95% confidence level will lead to this conclusion with respect to the suspect value: O Should be thrown away Should not be thrown away There is not enough data to draw a conclusion None of the above
Eight replicate measurements were made on a test solution producing the following results for the concentration of sodium ion (mg/L): 63.91, 65.92, 69.98, 64.24, 65.45, 65.82, 66.51, 65.11. The value 69.98 is an outlier suspect. A Q-test at 95% confidence level will lead to this conclusion with respect to the suspect value: O Should be thrown away Should not be thrown away There is not enough data to draw a conclusion None of the above
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.12QAP
Related questions
Question
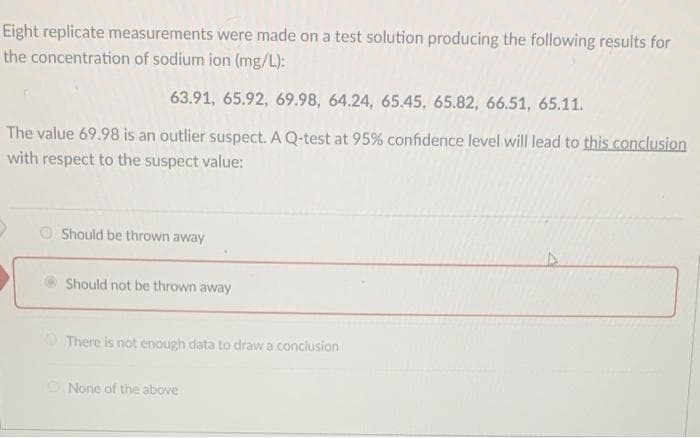
Transcribed Image Text:Eight replicate measurements were made on a test solution producing the following results for
the concentration of sodium ion (mg/L):
63.91, 65.92, 69.98, 64.24, 65.45, 65.82, 66.51, 65.11.
The value 69.98 is an outlier suspect. A Q-test at 95% confidence level will lead to this conclusion
with respect to the suspect value:
O Should be thrown away
Should not be thrown away
There is not enough data to draw a conclusion
None of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



